|
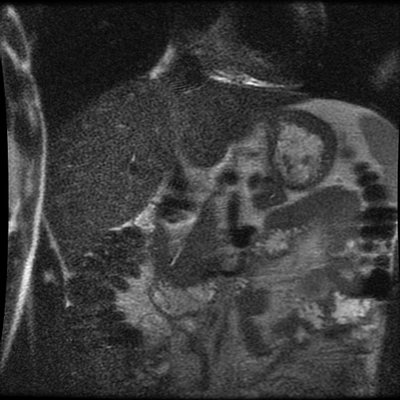
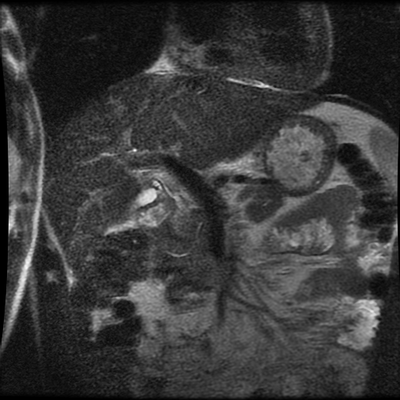
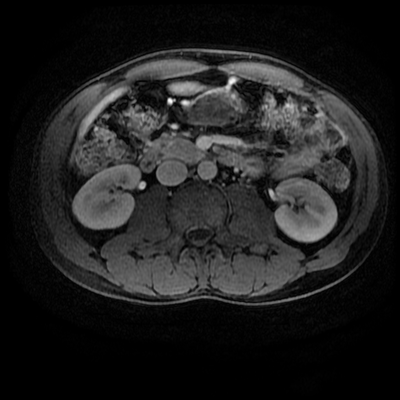
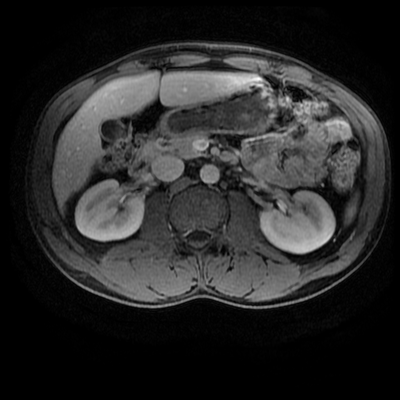
Level: MRCP, FRCR, FRCS, EDiR, Radiology Mid-Level ++ The images provided part of an MRCP acquisition. The two superior images are radial ray thick slab heavily T2-weighted acquisitions (oblique views). The second series of images are coronal thin section T2 weighted single shot fast spin-echo (SSFSE sequences). The third row of images are T1 fat suppressed volumetric acquisitions following intravenous gadolinium (LAVA, equivalent to VIBE or THRIVE). The final row of images are T2 axial images.
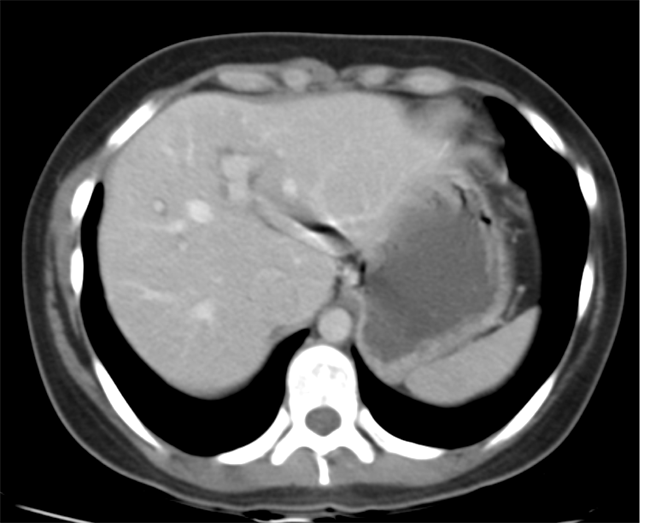
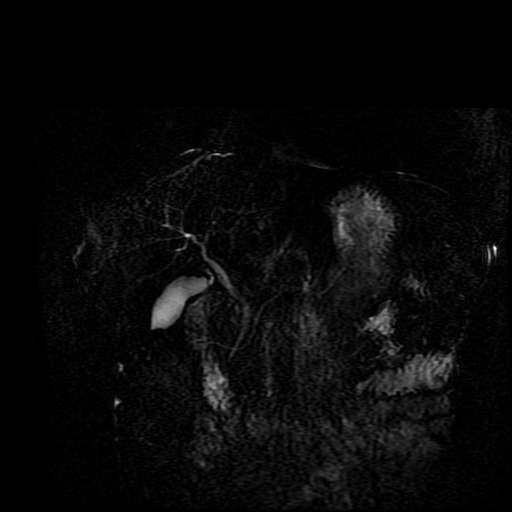
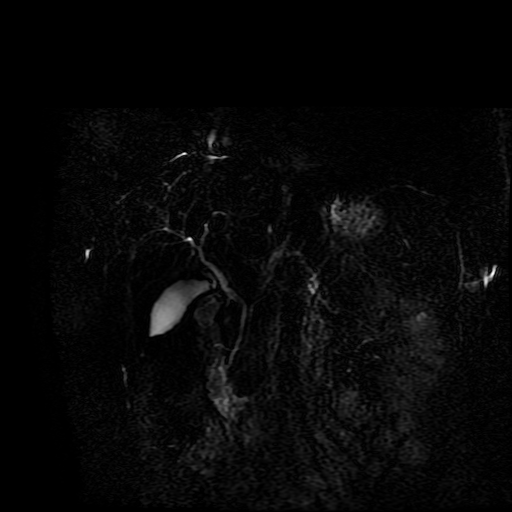
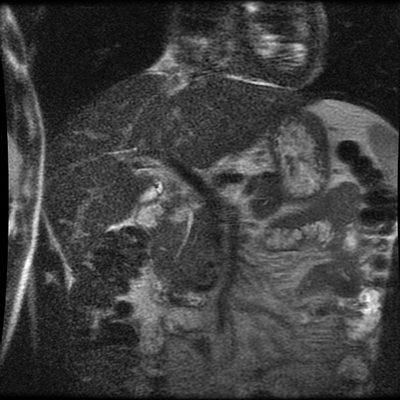
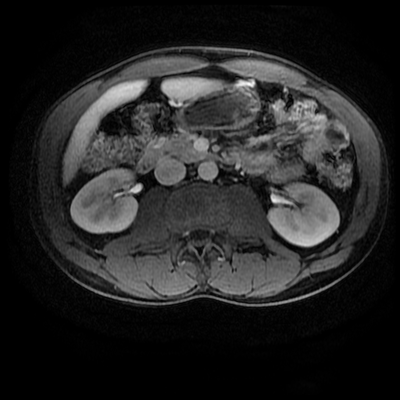
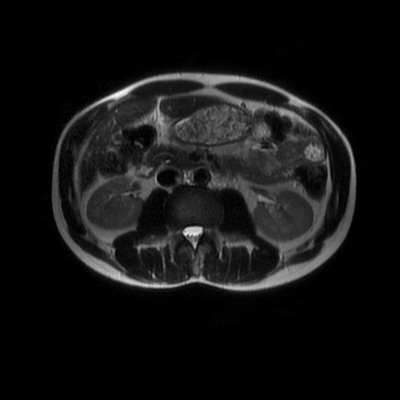
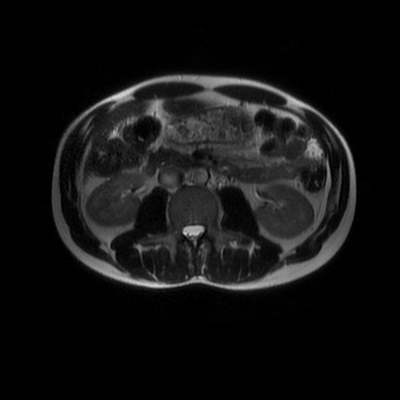
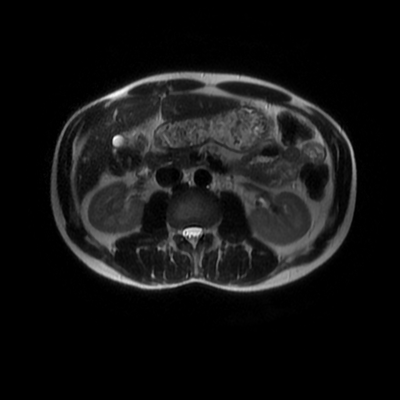
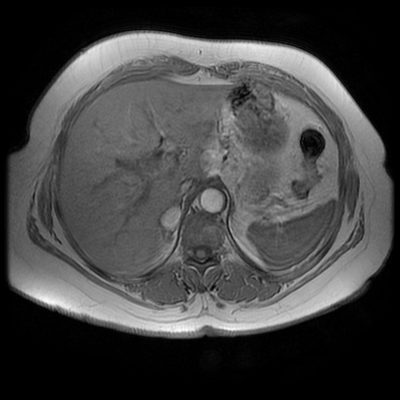
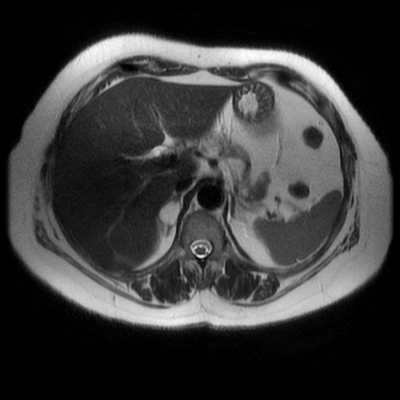
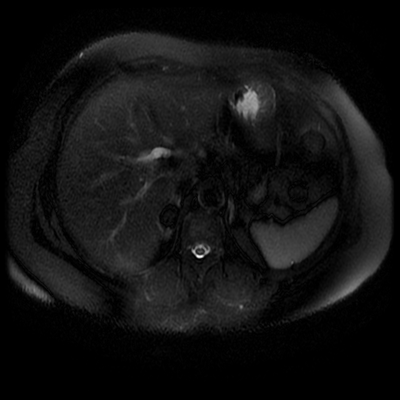
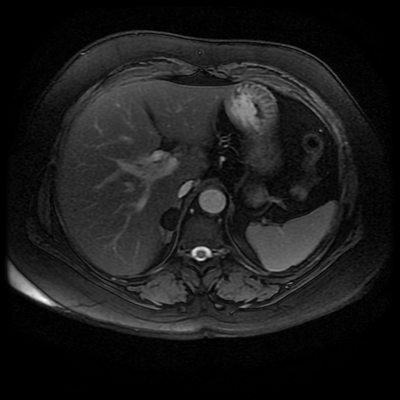
The images demonstrate that there is a crossover of the main pancreatic duct and of the distal common bile duct. The main pancreatic duct drains into the the accessory duct (Santorini) which emerges into the duodenum from the proximal minor papilla. The uncinate process of the pancreas drains via the main pancreatic duct (Wirsung) into the main ampulla, also receiving the common bile duct. These appearances are those of pancreas divisum. This appears due to failure of fusion of the ventral pancreatic bud with the larger dorsal pancreatic bud in the 8th week in utero. The ventral pancreas drains through the Wirsung duct which after the 8th week typically becomes the distal main pancreatic duct emerging into the duodenum at the ampulla. The duct of Santorini which drains the dorsal pancreas distally into the minor ampulla usually becomes atrophic allowing drainage through the more inferior Wirsung duct. Note that confusingly due to rotation of the pancreas in utero the posterior inferior head of the pancreas is actually congenitally the ventral pancreas. When the two duct systems fail to fuse pancreas divisum arises. Pancreas divisum is present in approximately 10% of patients, depending on how this is evaluated (postmortem 7%, ERCP 10-14%) and the definition of divisum. Although Asian populations have been reported to have lower incidence (1-2%) this has been stated in part in part to relate to less invasive testing. Many of the higher incidences of pancreas divisum relate to a definition that includes predominant flow into the duct of Santorini with small branch duct communication from the pancreatic duct to a small Wirsung duct. Radiologically the term at MRCP is usually confined to cases where there is complete separation with no bridging duct between the pancreatic duct in the neck/head and the Wirsung duct. This results in a crossover appearance of the CBD extending down into the ampulla and of the pancreatic duct extending into the duct of Santorini. Most cases of pancreas divisum are asymptomatic. In a small minority of cases recurrent pancreatitis may be related to pancreas divisum, due to inadequate drainage from the minor ampulla and the narrowed distal duct of Santorini. This can result in focal ovoid dilatation proximal to the ampulla (Santorinicoele). In these cases treatment can be by sphincterotomy or duct stenting. Level: FRCR, EDiR, ABR, Radiology Mid-Level ++ The images provided are:Top Right T1-weighted, top Left T2-weighted, bottom left T2 fat suppressed, bottom right FIESTA (T2 predominant gradient echo sequence with fat suppression). The imaging demonstrates a well defined ovoid lesion arising from the right adrenal gland. This lesion demonstrates diffuse high T1 and high T2 signal with diffuse suppression on the fat suppressed acquisitions.
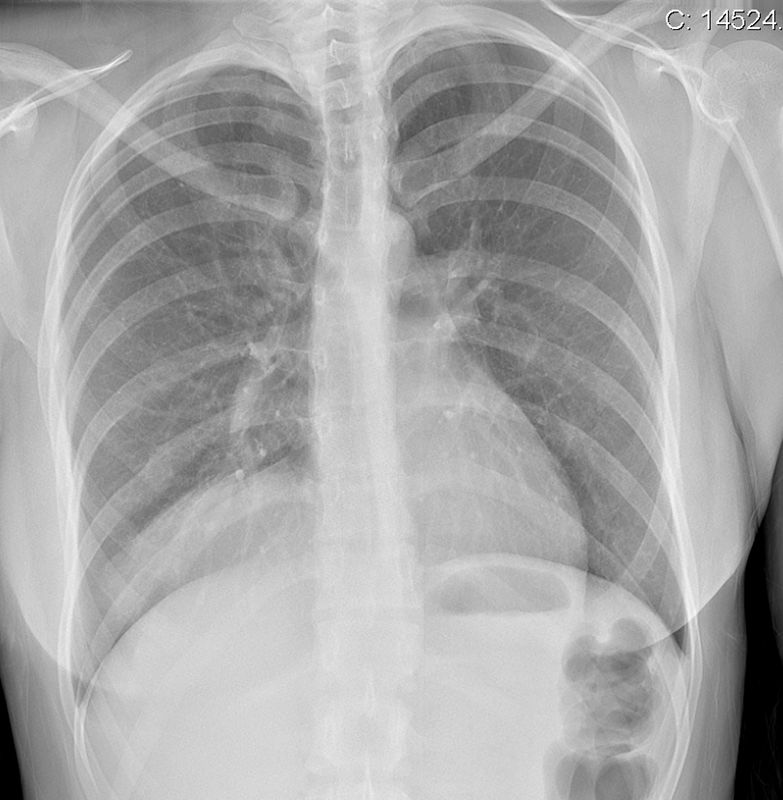
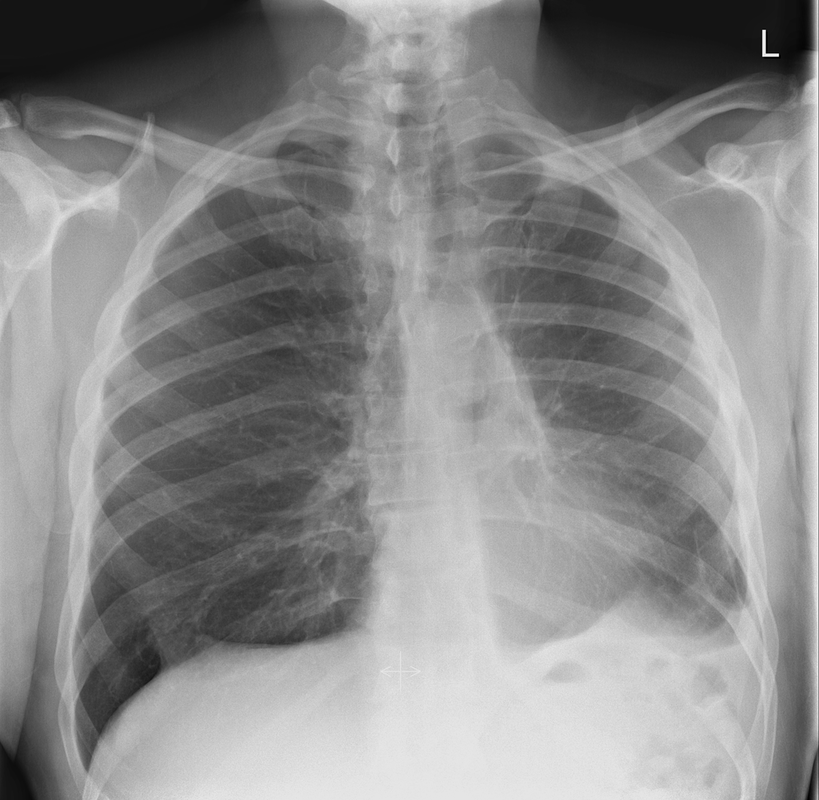
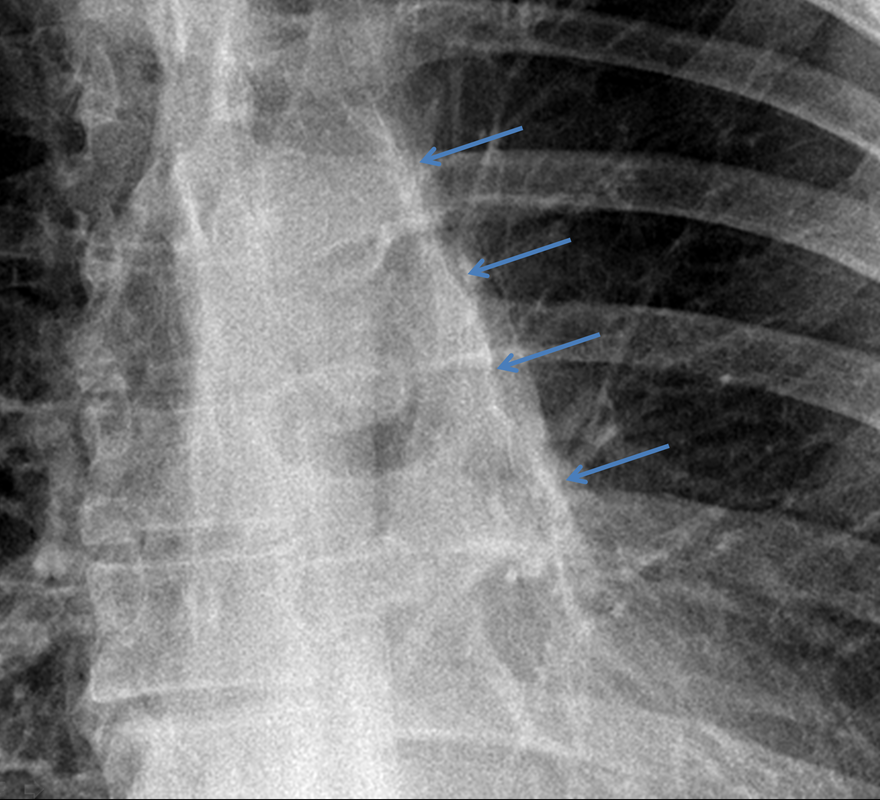
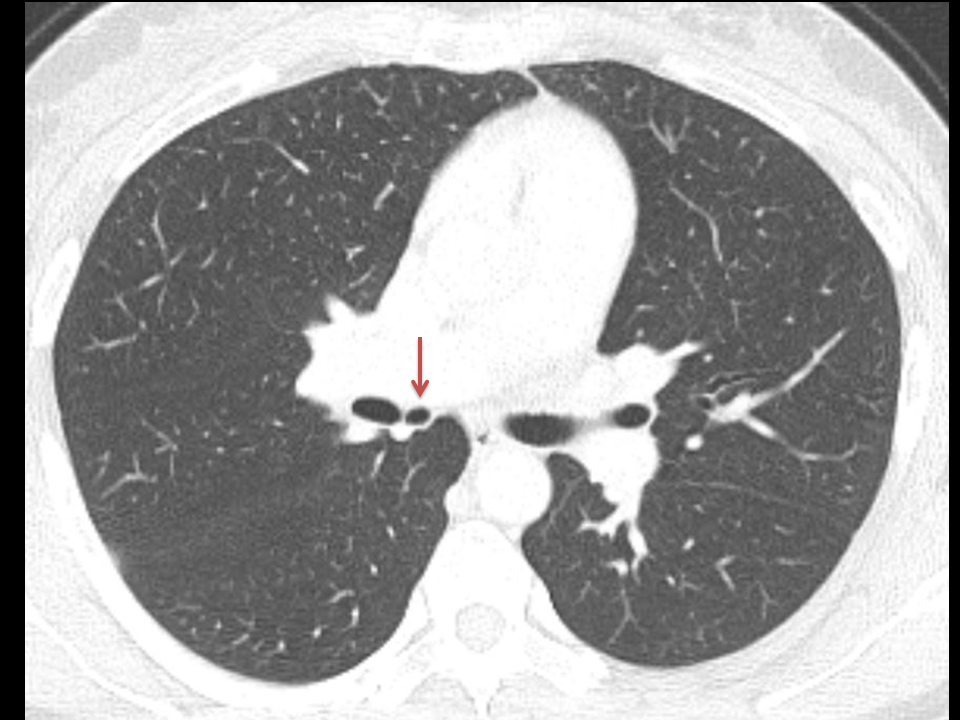
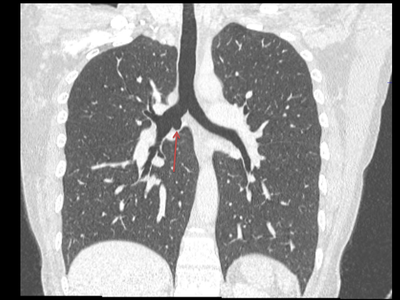
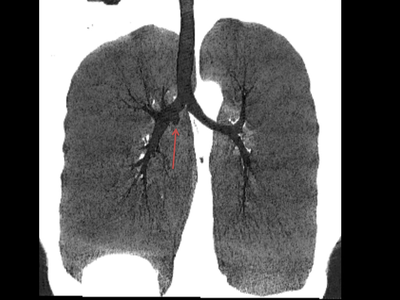
The commonest lesion of the adrenal gland is that of a benign adrenal adenoma, usually lipid-rich. This lipid is, however, microscopic fat. Microscopic fat can be equated to intra-voxel fat and is best demonstrated by paired T1 in and out of phase sequences, with loss of signal on the out of phase imaging. The demonstration of loss of signal uniformly in the right adrenal on these frequency encoded fat saturated images indicate the presence of bulk fat. Note that there is also suppression of the bulk fat in the peritoneum and subcutaneous tissues which is not demonstrated on typical T1 out of phase imaging. In this instance, therefore, the appearances are suggestive of a bulk (macroscopic) fat containing right adrenal lesion. This appearance is typically due to an adrenal myelolipoma. Adrenal myelolipomas are benign lesions, typically detected incidentally as in this case. The to the ill-defined or ill-defined. Occasionally when large there may be associated with a haemorrhagic presentation although this is rare and I have may be encountered asymptomatic lesions measuring greater than 5 or 10 cm. Occasional calcifications can be demonstrated. Adrenal myelolipomas are a composition of mature lipid laden adipocytes (lipoma) and some erythroid cells (myelo). They are themselves nonfunctioning although there are some occasional associations with other endocrine disorders such as Cushing's disease or Conn's syndrome. When diffusely fatty these lesions are easy to characterise and diagnose. There is a more complex consideration when these lesions contain significant soft tissue components as it is difficult differentiate the possibility of a collision tumour where a benign lesion such as a fat containing myelolipoma is incidentally adjacent to neoplastic lesion. Such lesions may require PET, follow-up or biopsy. The differential for bulk fat containing adrenal lesions is limited. There are rare described adrenal angiomyolipomas (comparable to renal angiomyolipoma). Other bulk fat containing lesions within the adrenal glands are also unusual and limited to liposarcoma or rarer fat containing adrenocortical carcinomas. Level: EDiR, ABR, FRCR Radiology Mid-Level ++ This case is a companion case to the Case 64, with many comparable features, many of which are more pronounced than the prior case. The left hemithorax is contracted with shift of both the superior and inferior mediastinum to the left. Comparable to the prior case there is also evidence of selective volume loss in the left upper thorax with elevation of the left hilar structures, a horizontal orientation of the left main bronchus and tenting of the left hemidiaphragm. However, in contradistinction to the prior case there is no perihilar opacity present. So this appears to reflect extreme left upper collapse but with no residual opacity visible to reflect the collapsed left upper lobe. There is an additional feature present. This is a fine line of high attenuation material running parallel to the mediastinal surface in the left perihilar region (arrow). The appearances are those of chain sutures as part of a left upper lobectomy which accounts for the volume loss differences demonstrated.
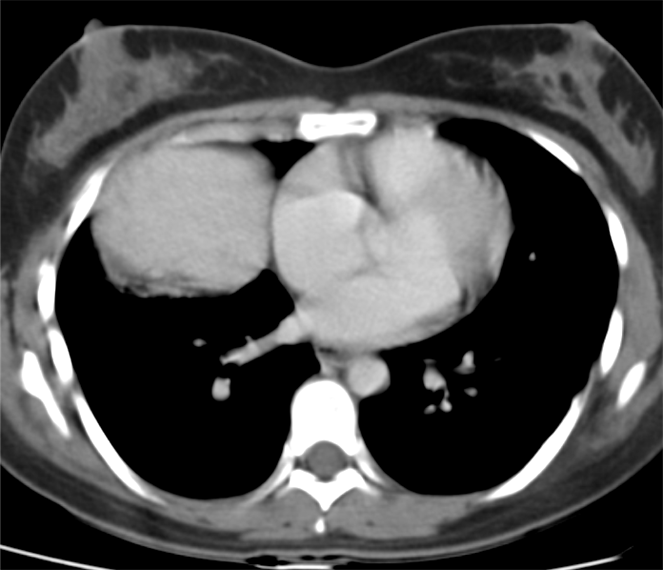
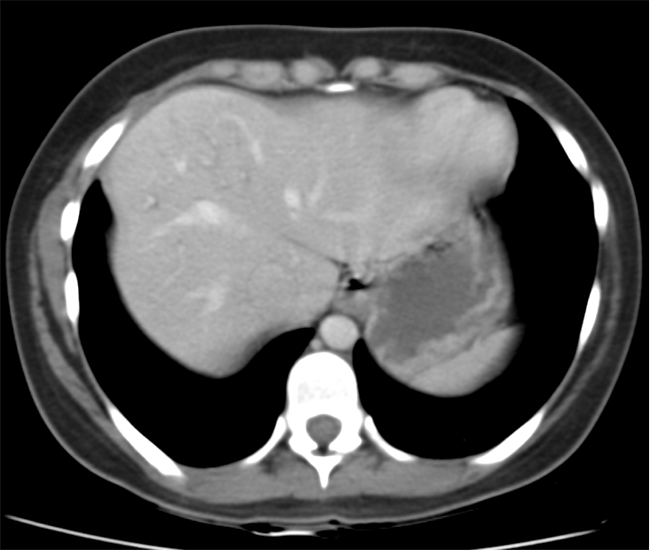
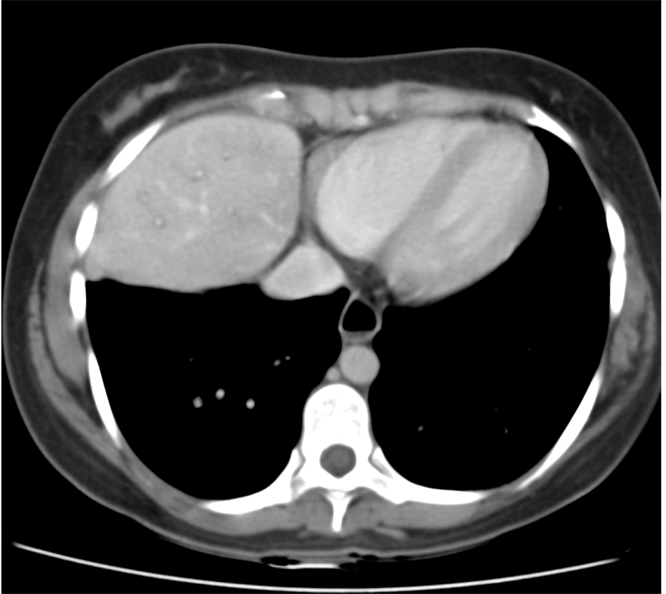
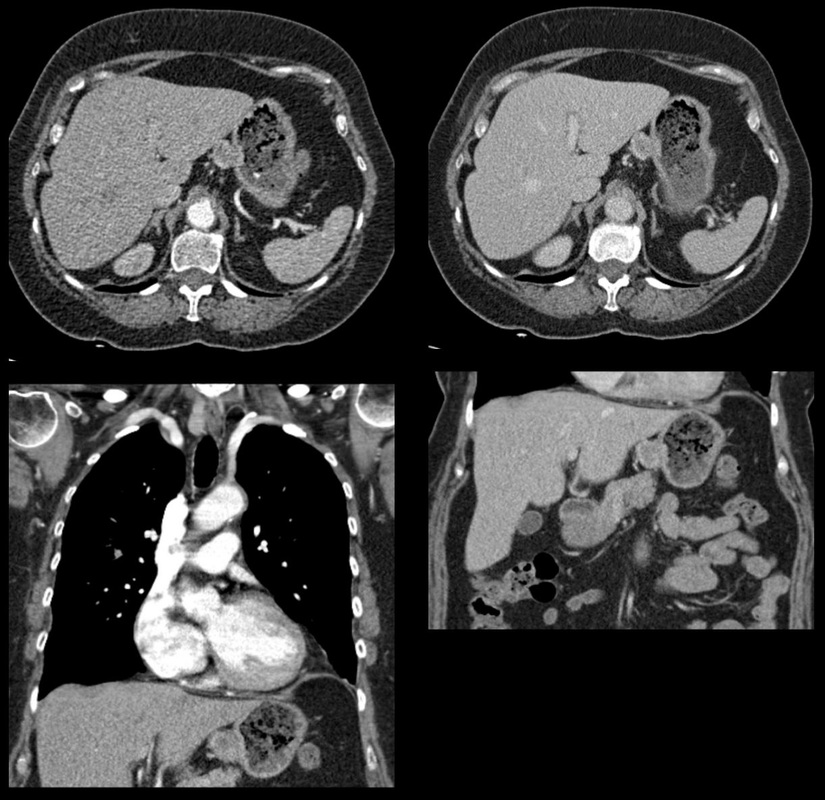
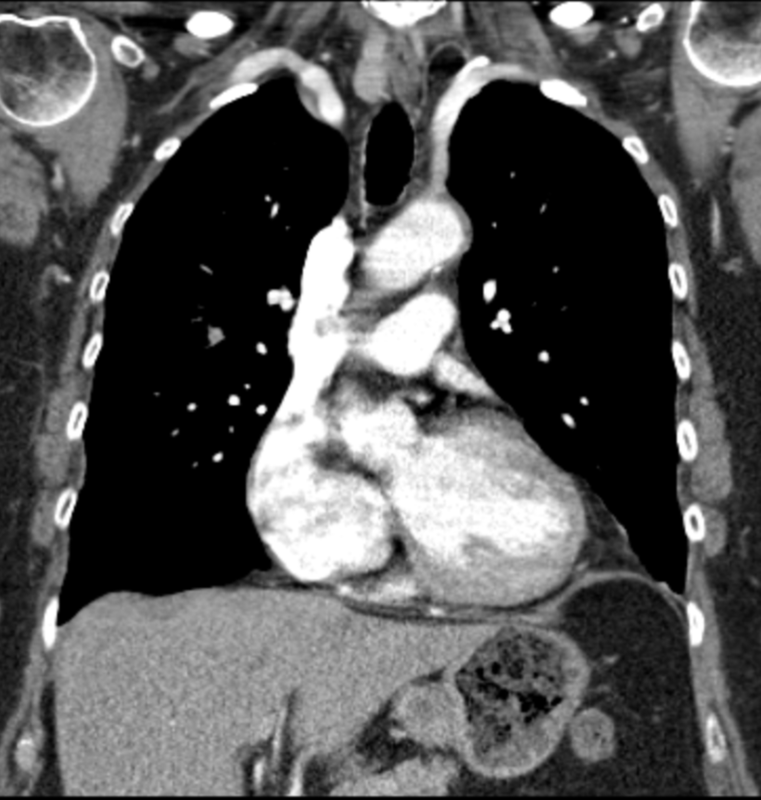
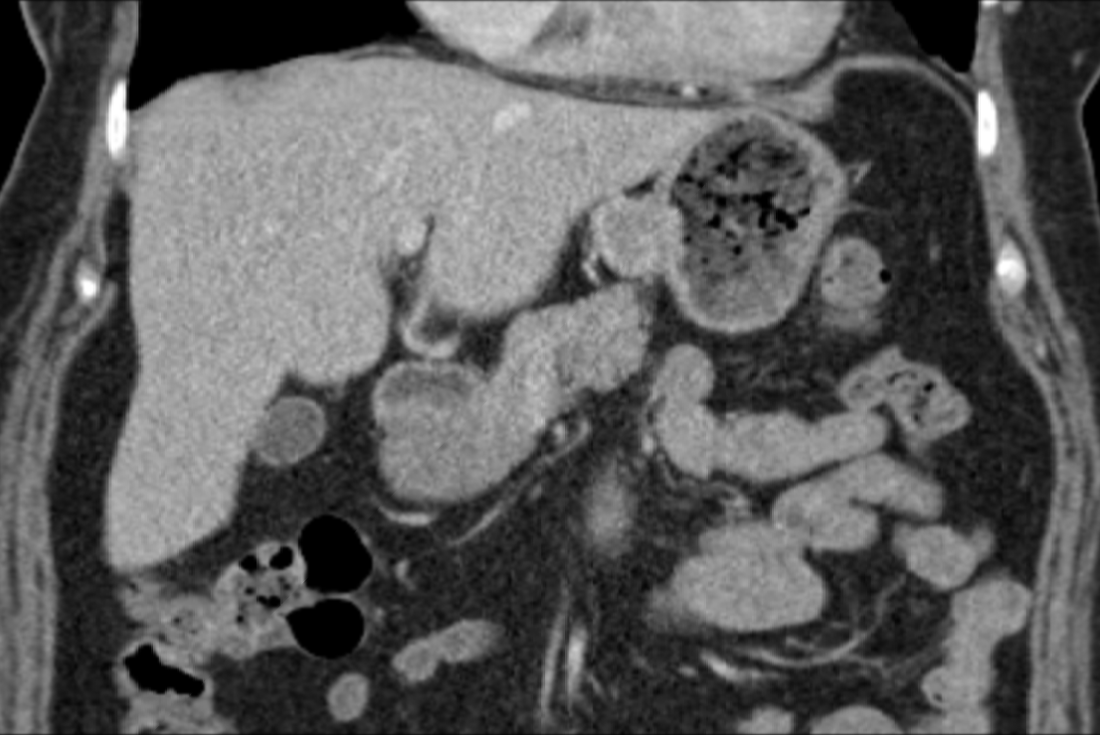
Lobectomy can be a difficult diagnosis to make. Appearances often mimic the characteristic appearances of lobar collapse but are lacking in the typical opacities that result from the collapsed lung parenchyma. Surgical clips are often conspicuous by their absence as these are rarely used or of a large enough size to be visualised on plain radiographs. Chain sutures can be variably used as in this case and be identified. These are used to complete an incomplete fissure and create a dividable plane so as to enable an air-leak free plane for an anatomical resection of the lobe. They can be difficult to see as they are so fine, resebling a very fine interlinked chain necklace. They are best visualised by magnification and sometime sharpening the image display. Post-operative changes of the ribs may also be appreciable, although this was more common in remote cases where a more extensive periosteal stripping was utilised that resulted in an irregular rib re- growth. In modern thoracotomy the rib irregularity is very focal and can be so subtle as to not even be identifiable even at CT. In post-lobectomy patients a small pleural effusion and persistent pleural thickening is often present at the lung bases, resulting in a long-standing pleural reaction. Determining the lobe of resection on plain radiographs relies on determining the position of the hilum and the central bronchovascular structures with expected superior displacement for upper lobe resections and conversely inferior displacement for lower lobe resections. Level: FRCR, ABR, EDiR Radiology Mid-Level ++ The images provided are thoracic arterial phase axial and coronal CT (left side) and abdominal portal venous phase axial and coronal CT (right side) post intravenous contrast. Both phases demonstrate that there is an exophytic lesion arising from the lesser curvature of the stomach. The lesion is peripherally enhancing and centrally heterogeneous. There is only minimal mucosal thickening of the gastric surface. No adenopathy is present.
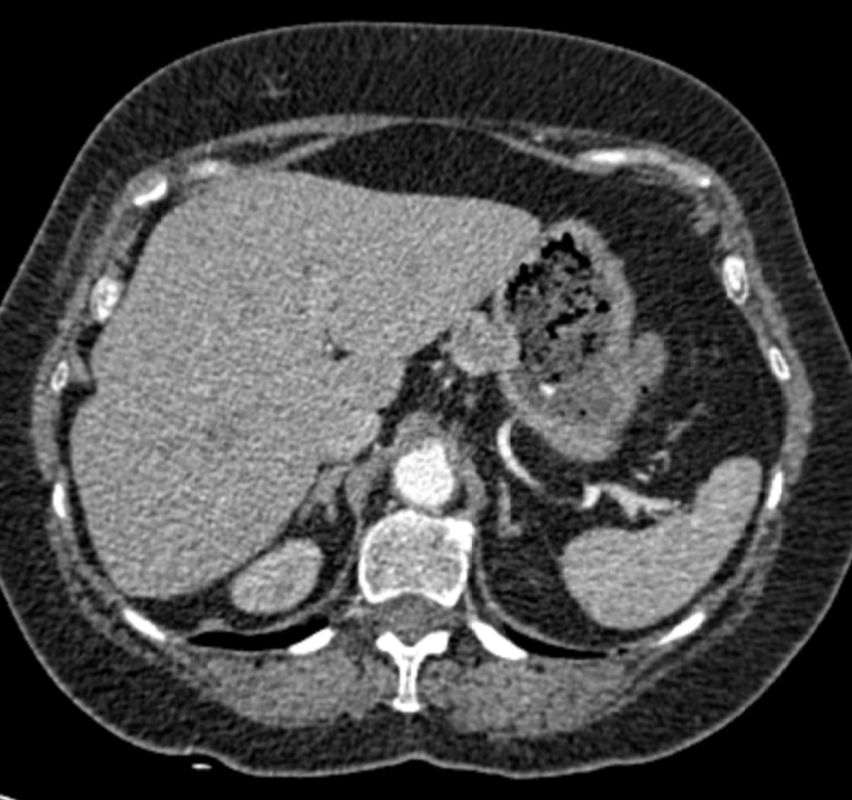
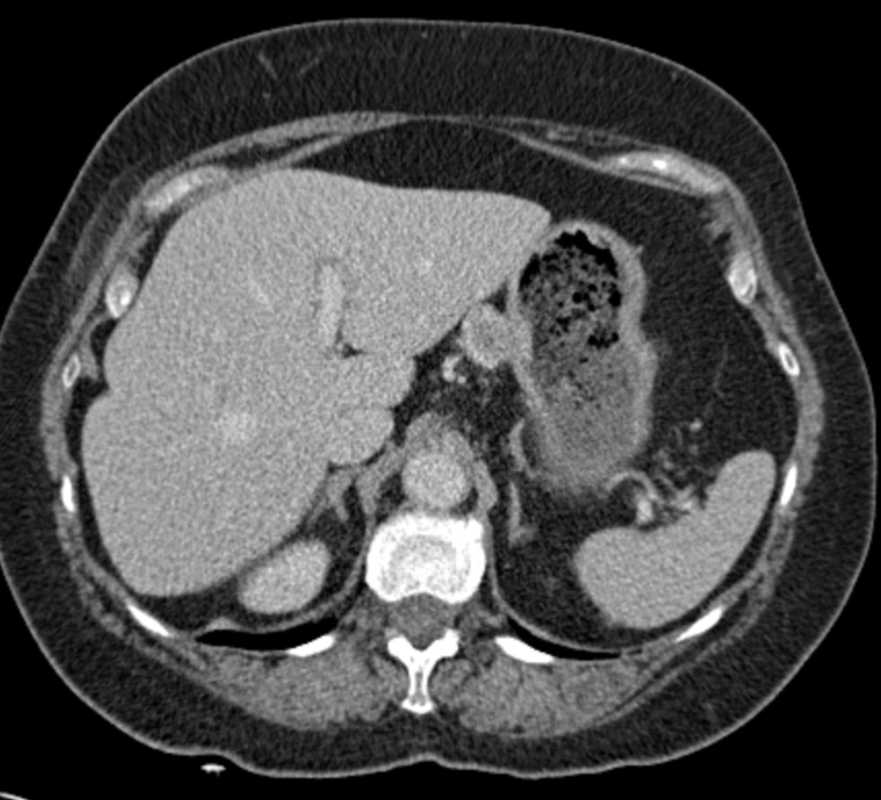
In this region the possibility of a node should be considered. Nodes in this region may reflect more widespread adenopathy, however, when isolated to this left gastric/gastrohepatic ligament region careful consideration of primary malignancies of the stomach or the lower oesophagus should be assessed. In this instance, however, the abnormality appears to arise from the gastric wall. These appearances are atypical for a gastric carcinoma that typically would present as an infiltrative or luminally narrowing lesion. The exophytic appearances in this case are typical of a gastrointestinal stromal tumour (GIST), a form of mesenchymal sarcoma, formerly known as leiomyoma or leiomyosarcoma. GISTs are most common in the stomach (70%) and are frequently exophytic either intra or less commonly extraluminally. They can reach a large size before presentation, particularly if extraluminal but not infiltrating into other structures. Ulceration, not evident in this case, occurs in up to half of lesions. This is visible in small lesions with barium studies or at CT in larger lesions, that may contain air from luminal extension. Haemorrhage is frequently present within the lesions and with central necrosis, as in this case, lead to tumour heterogeneous density or peripheral enhancement. GISTs are less frequently seen in other parts of the gastrointestinal tract, most commonly the small bowel, with lesser incidence in the oesophagus and elsewhere. Benign tumours of the oesophagus are still termed leiomyomas, where they are most common. GISTs can be entirely extra-enteric, termed extra-gastrointestinal occurring in the mesentery or retroperitoneum. The lesions arise from the submucosa so may be inconspicuous on endoscopy, or simply result in smooth indentation. Presentation is typically over the age of 40, unless there is a syndromic association, such as Carney’s triad (two or three of the following: pulmonary hamartomas, gastrointestinal stromal tumours and extra-adrenal phaeochromocytoma). Presentation can be incidental such as in this case, however, if intraluminal may present with obstructive symptoms or local symptomatology if locally infiltrative to other organs or metastatic (50%). Nodal metastases are uncommon. Gastrointestinal haemorrhage is a not infrequent cause of presentation. The presentation is in many ways linked to the aggressiveness of the tumour which is highly variable. Lesions <2cm are largely considered to have a benign progression and are unlikely to recur following resection. As distinct from genuine leiomyomas and leiomyosarcomas , GISTs contain no smooth muscle or Schwann cells. They are thought to arise from the interstitial cells of Cajal in the myenteric plexus. These cells normally express a trans-membrane receptor tyrosine kinase encoded by the KIT gene. The vast majority of GISTs exhibit activating mutations in KIT that lead to ligand-dependent KIT tyrosine kinases activation and promote tumor survival and growth. As a result treatment of irresectable GISTs has been greatly improved by the introduction of agents such as imatinib (Gleevec/Glivec) a tyrosine kinase inhibitor. From a radiological perspective it should be noted that treatment of GIST with imatinib may not result in a reduction in size of the tumour for several months. In addition early oedema or intratumoral haemorrhage (5%) may result in an early tumour transient increase in size. In addition to PET-CT SUV activity reduction, CT imaging can demonstrate reduction in attenuation of the tumour can be seen as early as one month following treatment initiation, and perhaps as early as one week. Choi has suggested modified response criteria for GIST treatment (10% decrease in unidimensional tumor size or a 15% decrease in tumour density on contrast-enhanced CT at 8 weeks post treatment) as a better correlation to PET-CT activity reduction. Fluid overload with ascites, pleural and pericardial effusions, or subcutaneous oedema is another effect of such therapy to be aware of. Additional Reading: Xie Hong et al. Gastrointestinal Stromal Tumor: Role of CT in Diagnosis and in Response Evaluation and Surveillance after Treatment with Imatinib http://pubs.rsna.org/doi/full/10.1148/rg.262055097 |
From Grayscale
Latest news about Grayscale Courses, Cases to Ponder and other info Categories
All
Archives
October 2018
|

|
|
Grayscale Courses est. 2015
 RSS Feed
RSS Feed
