|
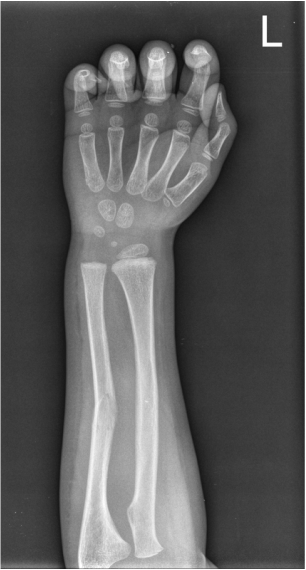
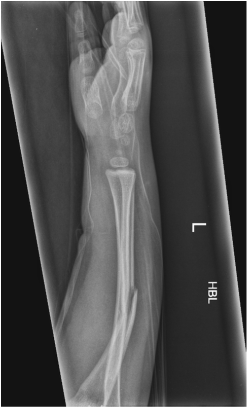
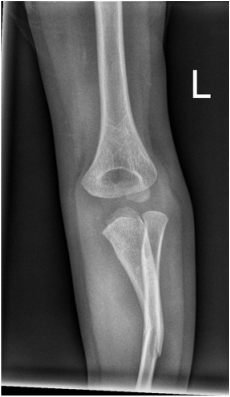
Case Level: Radiology Junior +, ABR, EDiR, MRCP, FRCR, FRCS The images demonstrate a relatively obvious fracture of the ulna. This is appreciable on the AP projection and confirmed as significantly angulated on the lateral projection. However, careful review of the elbow image reveals an additional abnormality. What is it?
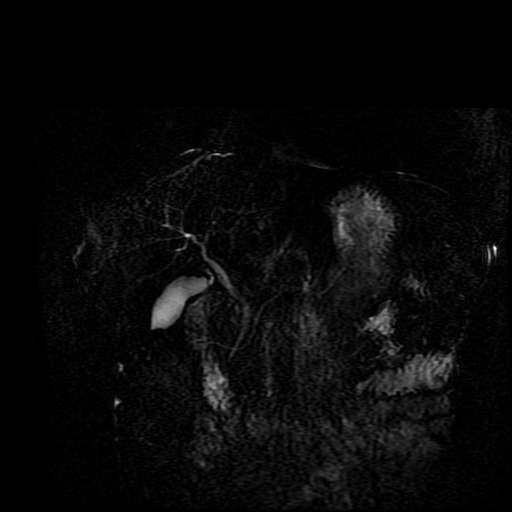
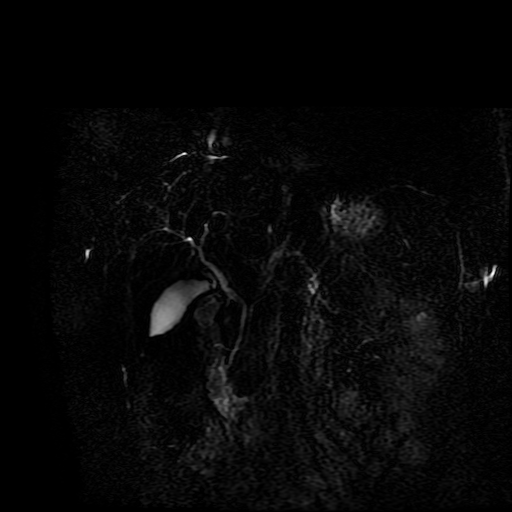
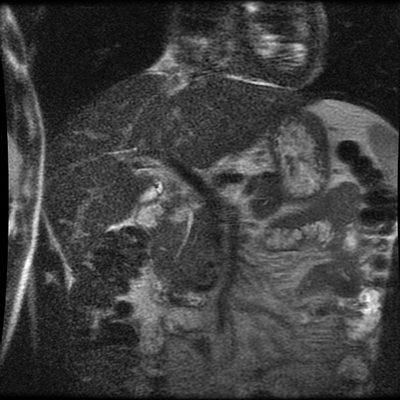
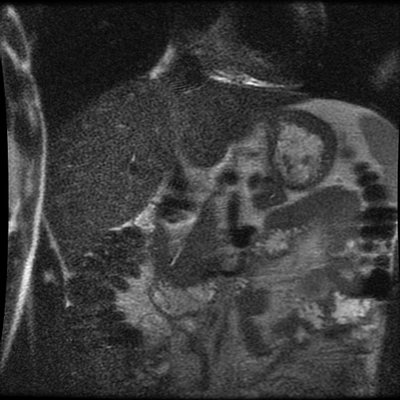
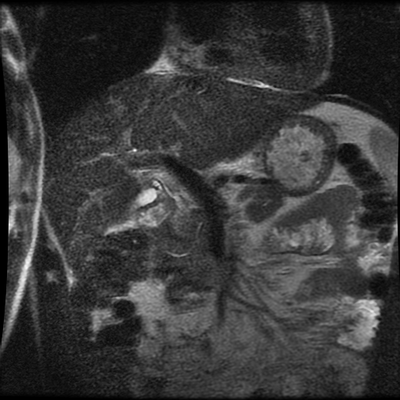
Whenever reviewing a paediatric elbow the first consideration is to establish that all the expected epiphyses are present. These appear in the order defined by the mnemonic of CRITOL (Capitellum, Radial head, Internal epicondyle, Trochlea, Olecranon, Lateral epicondyle). Epiphyses appearing out of order can be due to avulsion-dislocation of an epiphysis. In this case only the first epiphysis, the capitellum, in its correct position. However, the capitellum is the “cap” that lies over the radial head. In this case we can see that a line drawn through the medulla of the radius, through its expected head location does not meet the capitellum. Therefore, the findings reflect dislocation of the proximal radius/radial head. The combination of a mid-ulnar fracture and proximal radius/radial head dislocation constitutes the eponymous fracture-dislocation combination of Monteggia. This type of fracture dislocation highlights the importance of imaging the joint above and below a fracture. These types of fracture-dislocations are difficult for most to remember. The commonly confused fracture-dislocation is that of Galeazzi. In Galeazzi there is a mid-radial fracture with dislocation of the distal ulnar head at the wrist joint. It can be frustrating to remember the fracture and where the dislocation is (bone and location) and there are many mnemonics to assist. My preference is to recall that the dislocation occurs in the non-fractured bone and to use the mnemonic of GRIMUS (Galeazzi, Radius #, Inferior dislocation, Monteggia, Ulna #, Superior dislocation). Recall that the fracture may simply be a greenstick injury but it is essential to look for the associated dislocation as these can be missed and result in chronic pain/dysfunction for the patient and medicolegal pain for the reporting radiologist! Finally don’t forget the final fracture dislocation which is the Essex-Lopresti (one hyphenated British surgeon not 2 people!). This is a comminuted fracture of the radial head where force is transmitted down the interosseous ligament resulting in dissociation of the distal radio-ulnar joint at the wrist. Level: MRCP, FRCR, FRCS, EDiR, Radiology Mid-Level ++ The images provided part of an MRCP acquisition. The two superior images are radial ray thick slab heavily T2-weighted acquisitions (oblique views). The second series of images are coronal thin section T2 weighted single shot fast spin-echo (SSFSE sequences). The third row of images are T1 fat suppressed volumetric acquisitions following intravenous gadolinium (LAVA, equivalent to VIBE or THRIVE). The final row of images are T2 axial images.
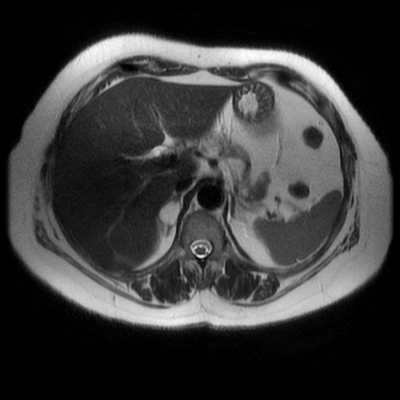
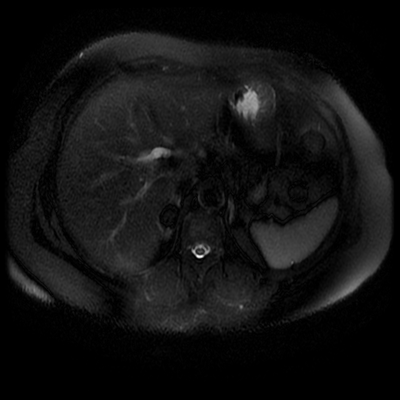
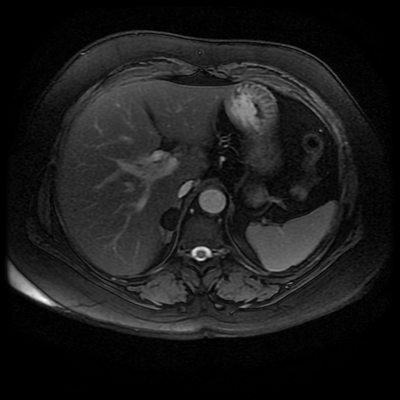
The images demonstrate that there is a crossover of the main pancreatic duct and of the distal common bile duct. The main pancreatic duct drains into the the accessory duct (Santorini) which emerges into the duodenum from the proximal minor papilla. The uncinate process of the pancreas drains via the main pancreatic duct (Wirsung) into the main ampulla, also receiving the common bile duct. These appearances are those of pancreas divisum. This appears due to failure of fusion of the ventral pancreatic bud with the larger dorsal pancreatic bud in the 8th week in utero. The ventral pancreas drains through the Wirsung duct which after the 8th week typically becomes the distal main pancreatic duct emerging into the duodenum at the ampulla. The duct of Santorini which drains the dorsal pancreas distally into the minor ampulla usually becomes atrophic allowing drainage through the more inferior Wirsung duct. Note that confusingly due to rotation of the pancreas in utero the posterior inferior head of the pancreas is actually congenitally the ventral pancreas. When the two duct systems fail to fuse pancreas divisum arises. Pancreas divisum is present in approximately 10% of patients, depending on how this is evaluated (postmortem 7%, ERCP 10-14%) and the definition of divisum. Although Asian populations have been reported to have lower incidence (1-2%) this has been stated in part in part to relate to less invasive testing. Many of the higher incidences of pancreas divisum relate to a definition that includes predominant flow into the duct of Santorini with small branch duct communication from the pancreatic duct to a small Wirsung duct. Radiologically the term at MRCP is usually confined to cases where there is complete separation with no bridging duct between the pancreatic duct in the neck/head and the Wirsung duct. This results in a crossover appearance of the CBD extending down into the ampulla and of the pancreatic duct extending into the duct of Santorini. Most cases of pancreas divisum are asymptomatic. In a small minority of cases recurrent pancreatitis may be related to pancreas divisum, due to inadequate drainage from the minor ampulla and the narrowed distal duct of Santorini. This can result in focal ovoid dilatation proximal to the ampulla (Santorinicoele). In these cases treatment can be by sphincterotomy or duct stenting. Level: ABR, EDiR, FRCR, Radiology Challenge ++++ The first set of images are 2-D time-of-flight non contrast MRI acquisitions through the aortic arch and root of the neck. The second set of images are coronal MRA acquisitions of the same region following intravenous gadolinium administration. The final image is the maximum intensity projection of the MRA acquisition.
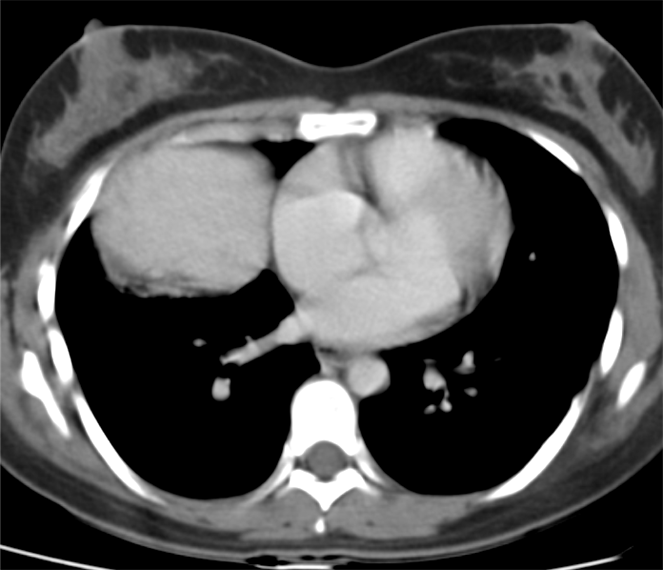
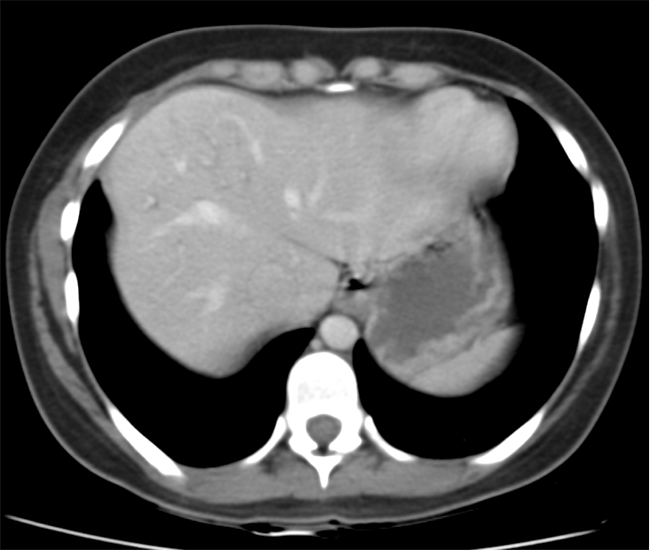
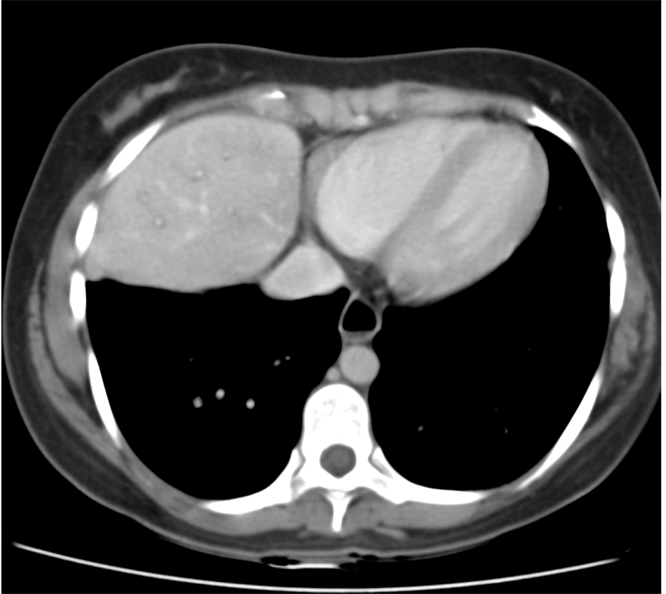
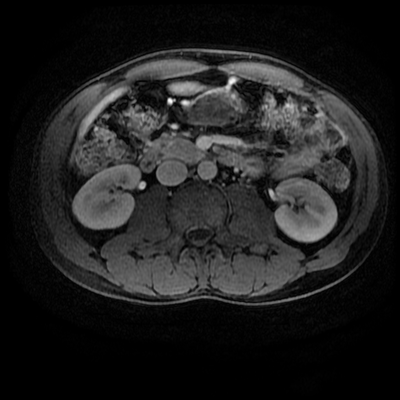
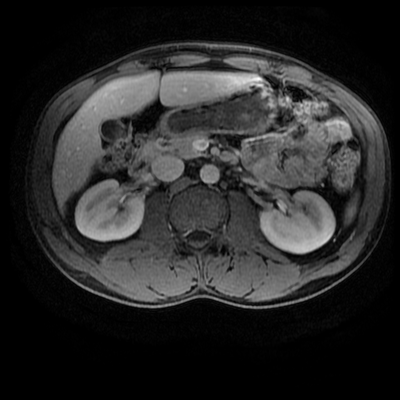
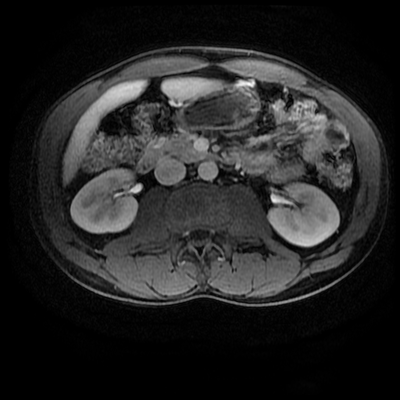
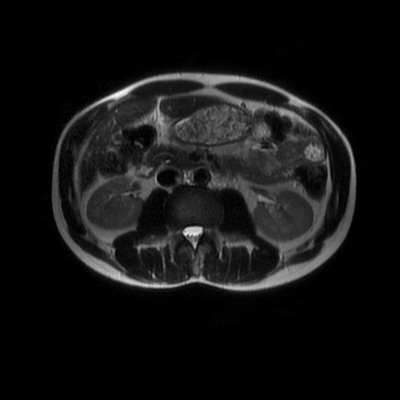
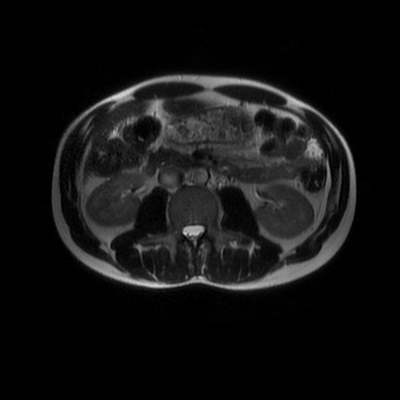
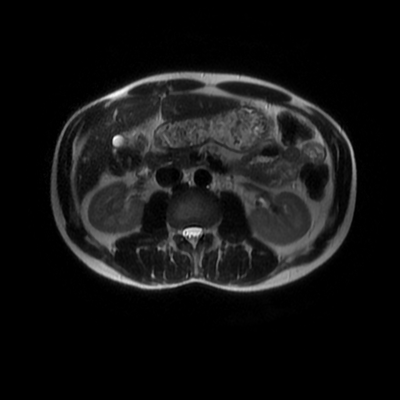
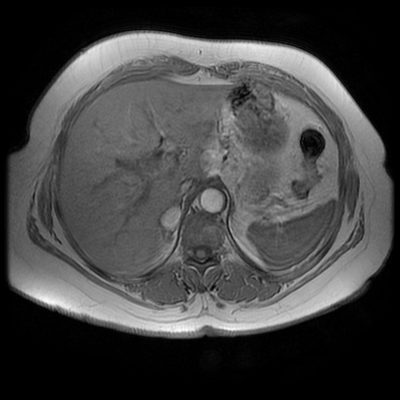
Both series of images demonstrate a large vessel arising from the posterior aspect of the aortic arch, running to the right side posterior to the oesophagus and then cranial to the right supraclavicular region. This is an incidental left-sided aortic arch with an aberrant right subclavian artery configuration. The aberrant right subclavian artery is arteriomegalic. Technically this is not a diverticulum of Kommerrel, from which these can arise as a focal aneurysmal dilatation of the origin, but rather a vessel that is enlarged throughout its length. Patients with a left -sided aortic arch and aberrant right subclavian artery do not have a complete vascular ring, however, as in this case, may have compression of the oesophagus in this region, sometimes referred to as "dysphagia lusoria". One would expect that with the aberrant right subclavian artery instead of a conventional brachiocephalic artery there would be 4 vessels arising from the aortic arch (in turn: right common carotid, left common carotid, left subclavian, aberrant right subclavian). There are indeed 4 vessels arising off the aortic arch on the 2-D time-of-flight images. However, on the left side both extend into the neck. There is indeed no flow within the left subclavian artery at any level. What is the fourth vessel? This is a direct origin of the left vertebral artery from the aorta. This occurs in approximately 5% in patients instead of origin from the proximal left subclavian artery. However, the MRA contrast enhanced images, including the maximum intensity projection, do demonstrate enhancement of 5 arch vessels including the more peripheral left subclavian artery with a more proximal occlusion of this vessel, over approximately 1.5 cm. There are asymmetric tortuous collateral vessels in the left side of the neck collateralising to the subclavian artery. How can we account for the lack of left subclavian artery visualisation on the 2D time of flight sequence but visualisation, albeit incomplete, on the MRA study? What does this tell us? Well the 2-D time-of-flight images are important as they indicate directionality of flow. Note that only cephalad directed arterial flow is recorded. As every slice is acquired sequentially, a saturation pulse is sequentially applied above the next imaged slice to eliminate in-flow signal from structures flowing from above the slice into the slice at the time of acquisition (ie venous blood), allowing only in-flow signal from flowing structures below the slice (arterial). If we wanted insted to visualise venous flow without arterial contamination we would place the saturation pulse below every slice in the 2D time of flight. So this indicates that there is no cephalad flow in the left subclavian vein. But since there is opacifaction on the contrast enhanced MRA acquisition, the vessel must be partly patent. However, the flow must be retrograde (ie caudally directed). This is a not uncommon situation in vertebral steal phenomenon. A stenosis of the origin of the left subclavian results in poor left subclavian arterial perfusion. During periods of increased left subclavian arterial demand (exercise), the left subclavian artery fills retrogradely via the left vertebral artery (steal) which anastomoses across the circle of Willis and then connects to the subclavian artery distal to the obstruction. In this case, however, as the vertebral artery arises directly of the aorta at this collateral pathway is not available. We can see on the MRA examination that there is an additional tortuous vessel in the left neck anastomosing distally to the left subclavian artery. This vessel is not visualised on the 2-D time-of-flight acquisitions, indicating that this is downard flow supplying the distal left subclavian artery. Therefore, in combination these findings indicate that there is only retrograde filling of the left subclavian artery from these left-sided cervical branches. This is comparable to subclavian steal, but because of separate origin of the vertebral artery the retrograde filling of the left subclavian is not occurring from the vertebral itself. There is a complex case with two congenital variations of which one, the left vertebral direct aortic origin, is clinically pertinent. Analyzing the difference between 2-D time-of-flight and MRA images assists in determining directionality of flow, and in this case clinical physiology. Level: FRCR, EDiR, ABR, Radiology Mid-Level ++ The images provided are:Top Right T1-weighted, top Left T2-weighted, bottom left T2 fat suppressed, bottom right FIESTA (T2 predominant gradient echo sequence with fat suppression). The imaging demonstrates a well defined ovoid lesion arising from the right adrenal gland. This lesion demonstrates diffuse high T1 and high T2 signal with diffuse suppression on the fat suppressed acquisitions.
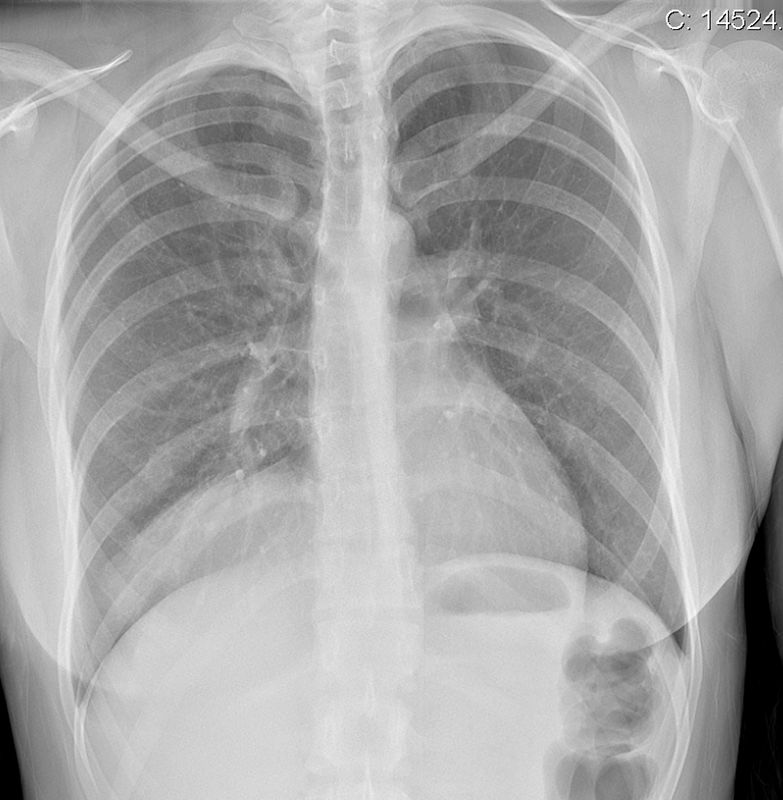
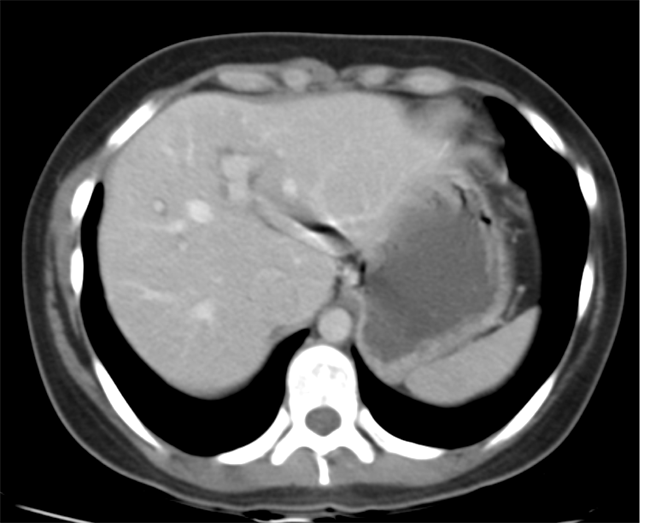
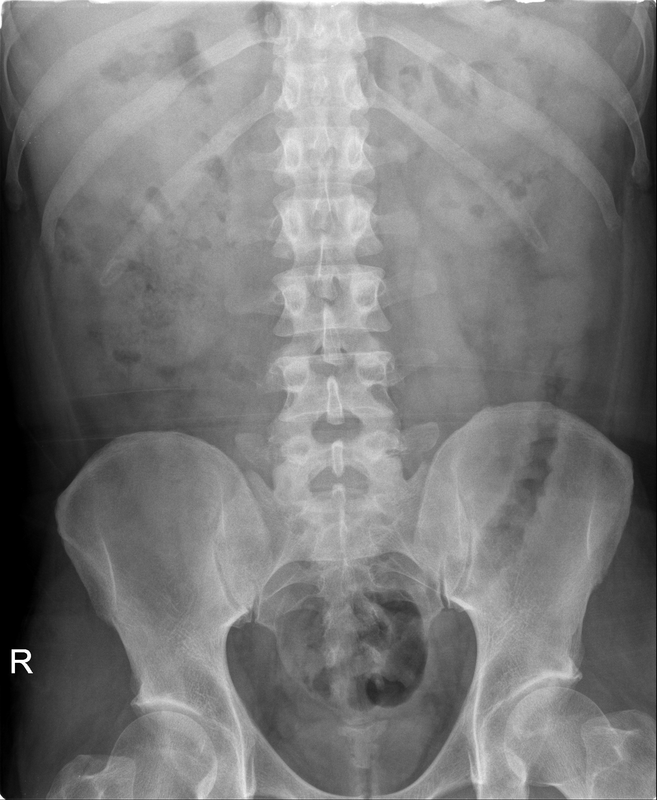
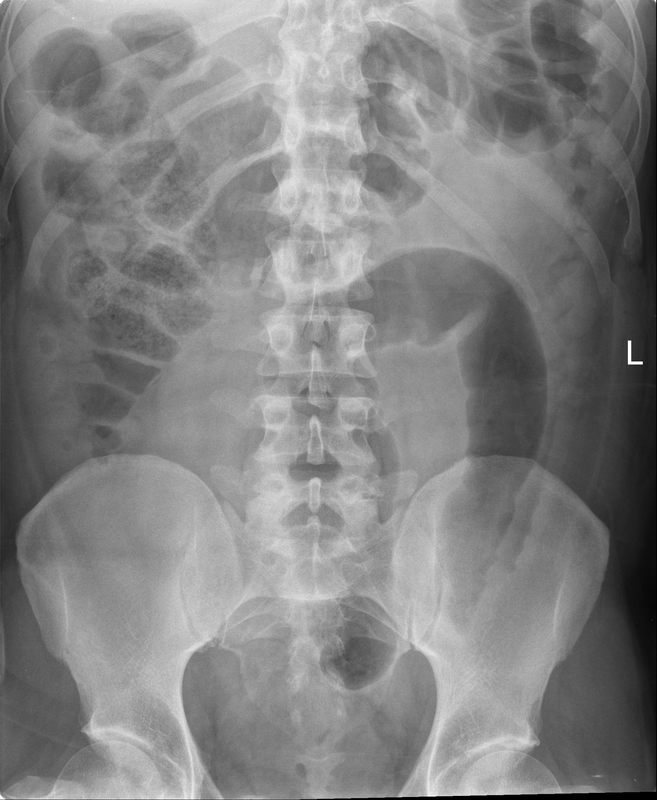
The commonest lesion of the adrenal gland is that of a benign adrenal adenoma, usually lipid-rich. This lipid is, however, microscopic fat. Microscopic fat can be equated to intra-voxel fat and is best demonstrated by paired T1 in and out of phase sequences, with loss of signal on the out of phase imaging. The demonstration of loss of signal uniformly in the right adrenal on these frequency encoded fat saturated images indicate the presence of bulk fat. Note that there is also suppression of the bulk fat in the peritoneum and subcutaneous tissues which is not demonstrated on typical T1 out of phase imaging. In this instance, therefore, the appearances are suggestive of a bulk (macroscopic) fat containing right adrenal lesion. This appearance is typically due to an adrenal myelolipoma. Adrenal myelolipomas are benign lesions, typically detected incidentally as in this case. The to the ill-defined or ill-defined. Occasionally when large there may be associated with a haemorrhagic presentation although this is rare and I have may be encountered asymptomatic lesions measuring greater than 5 or 10 cm. Occasional calcifications can be demonstrated. Adrenal myelolipomas are a composition of mature lipid laden adipocytes (lipoma) and some erythroid cells (myelo). They are themselves nonfunctioning although there are some occasional associations with other endocrine disorders such as Cushing's disease or Conn's syndrome. When diffusely fatty these lesions are easy to characterise and diagnose. There is a more complex consideration when these lesions contain significant soft tissue components as it is difficult differentiate the possibility of a collision tumour where a benign lesion such as a fat containing myelolipoma is incidentally adjacent to neoplastic lesion. Such lesions may require PET, follow-up or biopsy. The differential for bulk fat containing adrenal lesions is limited. There are rare described adrenal angiomyolipomas (comparable to renal angiomyolipoma). Other bulk fat containing lesions within the adrenal glands are also unusual and limited to liposarcoma or rarer fat containing adrenocortical carcinomas. Level: MRCP, FRCS, ABR, EDiR, FRCR, Radiology Level Junior + The initial image demonstrates a plain abdominal radiograph. Superficially the radiograph appears normal with no evidence of abnormal calcification projected over the kidneys, gallbladder or pelvis. There is no overt bowel dilatation to suggest obstruction. No free air is appreciated. Subtle abnormality is, however, demonstrable in the left flank where the descending colon extending into the sigmoid demonstrates thickening, characterised by indentations of the lateral and to a lesser degree the medial surface of the bowel. The properitoneal fat plane, the thin lateral lucency, which differentiates the descending colon from the left abdominal paracolic gutter is widened supporting bowel wall thickening in this region. These findings are suggestive of an early colitis.
The following radiograph performed 3 days later demonstrates that there is now dilatation of the distal descending colon and sigmoid. The thickening of the distal sigmoid and of the proximal aspect of the sigmoid can be noted overlapping over the left iliac wing. There is now more significant proximal small bowel dilatation which is likely due to ileus. These appearances are suggestive of a colitis. Initial mural thickening, often termed "thumbprinting" as in this case, reflect bowel wall oedema. It is then followed by bowel atony and gaseous distended ileus. Many similar etiologies of colitis can result in similar appearance including inflammatory bowel disease (usually ulcerative colitis), but also pseudomembranous or other infectious colitis. In this case the disease process was due to ulcerative colitis and serial daily imaging was performed to assess the possibility of a toxic megacolon. Maximal dilatation was at the 3 day film. Toxic megacolon, typically affects the transverse colon which may develop pronounced lung printing, loss of haustration and formation of inflammatory polyps. As the colon distends to 6 cm in greater there is a perforation is significantly increased. Perforation clearly is associated with high morbidity and mortality. |
From Grayscale
Latest news about Grayscale Courses, Cases to Ponder and other info Categories
All
Archives
October 2018
|

|
|
Grayscale Courses est. 2015
 RSS Feed
RSS Feed
