|
Radiology Level: FRCR, FRCS, MRCP, ABR, EDiR, Radiology Senior +++
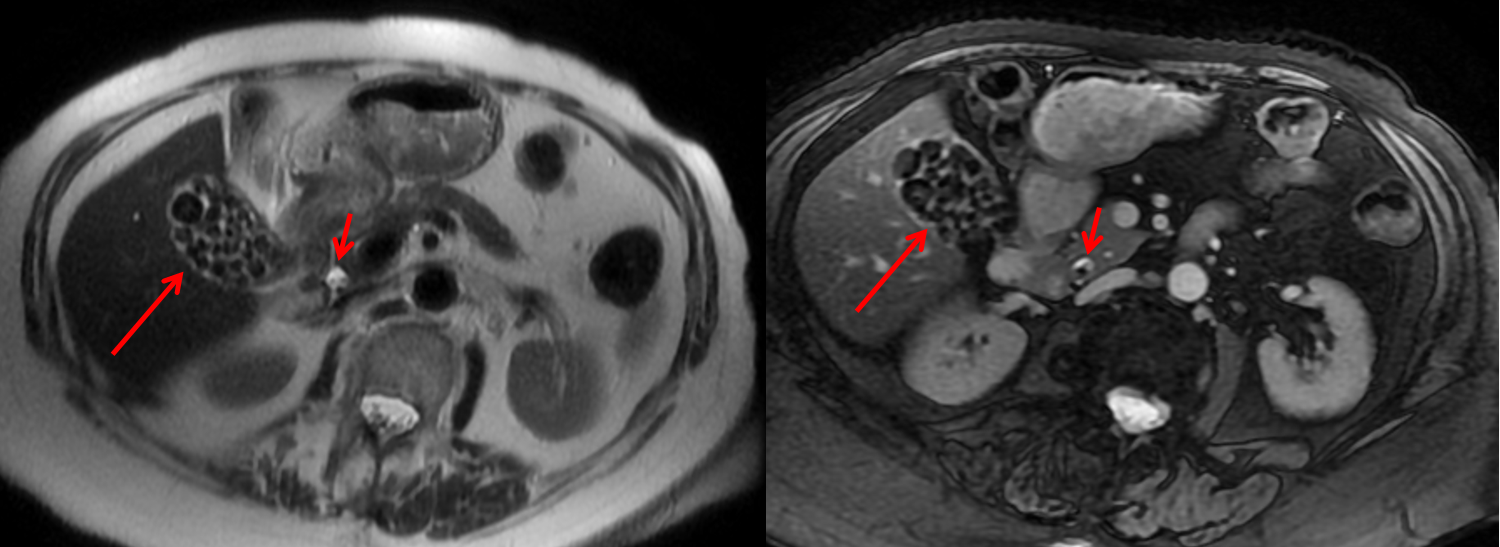
An interesting case to analyse. The abdominal radiograph demonstrates extensive areas of intrabdominal calcification. These do not conform to any intrabdominal organ, nor the bowel to suggest bowel contents. Therefore, these findings are likely peritoneal in nature. There are not many causes of peritoneal calcification. Intuitively, tuberculosis and peritoneal mesothelioma may come to mind. However, in the former calcification is unusual and limited and localised when present. Calcification is even rarer within peritoneal mesothelioma even if pleural plaques are calcified in the same patient. So it is important to consider other conditions. Pseudomyxoma peritonei can be associated with small areas of focal punctate or linear calcification. In these cases the bowel would be expected to be centrally displaced by the gelatinous soft tissue filling the peritoneum. This is not present in this case. Two conditions should be considered with extensive calcifications of this type. These are psammomatous calcifications in ovarian malignancy and the calcifications of sclerosing peritonitis. These two types of calcifications can be differentiated by their morphology. Psammoma bodies are usually fluffy calcifications that appear almost to be forming ossified bodies. In distinction the calcfications of sclerosing peritonitis are usually linear or sheet like. Therefore, in this case the calcifications are typical of sclerosing (or encapsulated) peritonitis. This is a calcification pattern that develops in patients on peritoneal dialysis. The appearances are often referred to as a "cocoon abdomen". The presence of the calcifications and peritoneal loculations impairs the ability to perform peritoneal dialysis effectively by reducing the peritoneal surface accessible to the dialysate fluid. When identifying features of one disease it is also important to look for further features that may corroborate the process. What about the bones, are they normal? The bone density is diffusely increased, the trabeculae are ill-defined, there is alternate sclerosis and lucency of the vertebral bodies ("rugger jersey spine"). These are features of renal osteodystrophy. The sacro-iliac joints are also ill-defined and widened. This is a feature of the hyperparathyroidism component of renal osteodystrophy resulting in subperiosteal bone resorption. Finally, vascular calcifications are present. These are less specific and seen not only in renal impairment but also in diabetes and elderly patients with atherosclerosis. The surgical clips in the pelvis are not related to a prior transplant as suggested by some, but rather due to sterilisation clips. Unrelated clips in the upper abdomen are due to unrelated surgery. Always look at all elements of a case. The bones, the soft tissues, the gas pattern, they may all assist in confirming a diagnosis and finding all its manifestations. Radiology Level: FRCR, FRCS, MRCP, ABR, EDiR, Radiology Challenge ++++
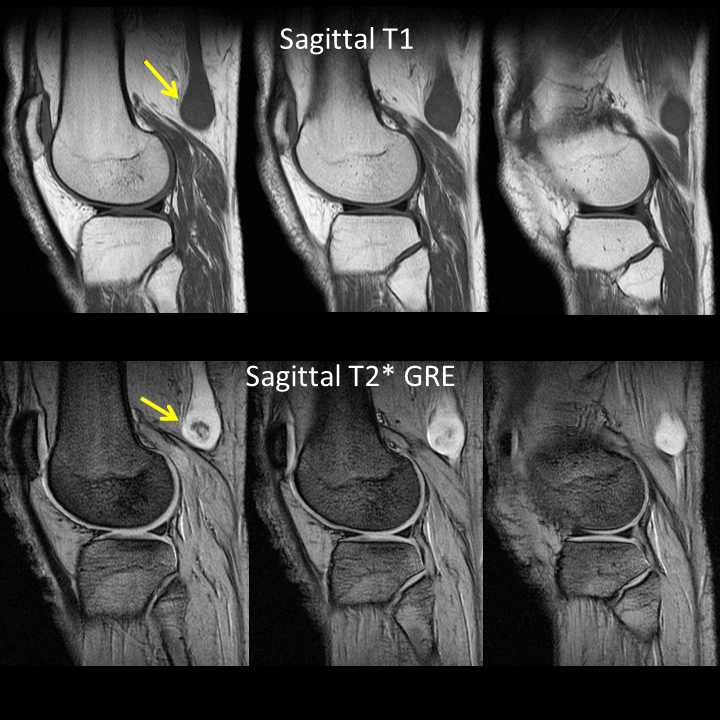
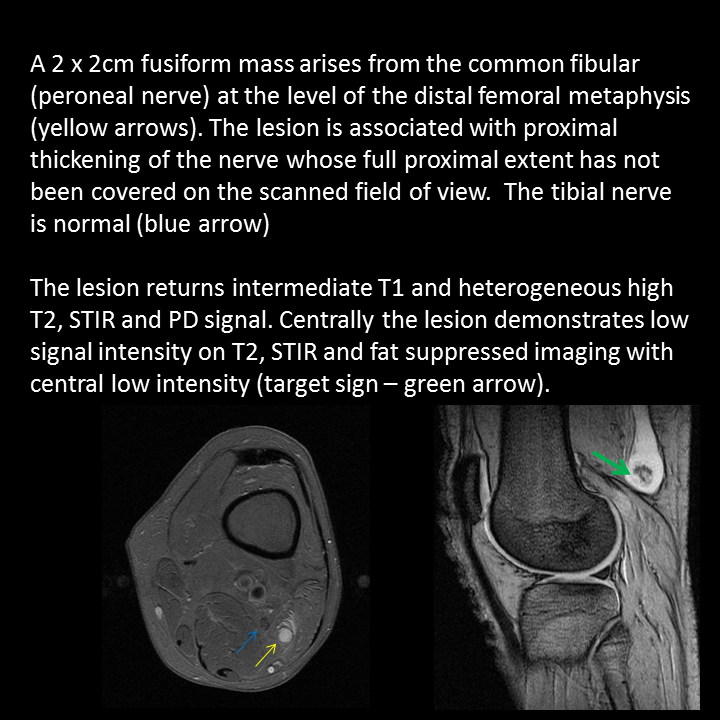
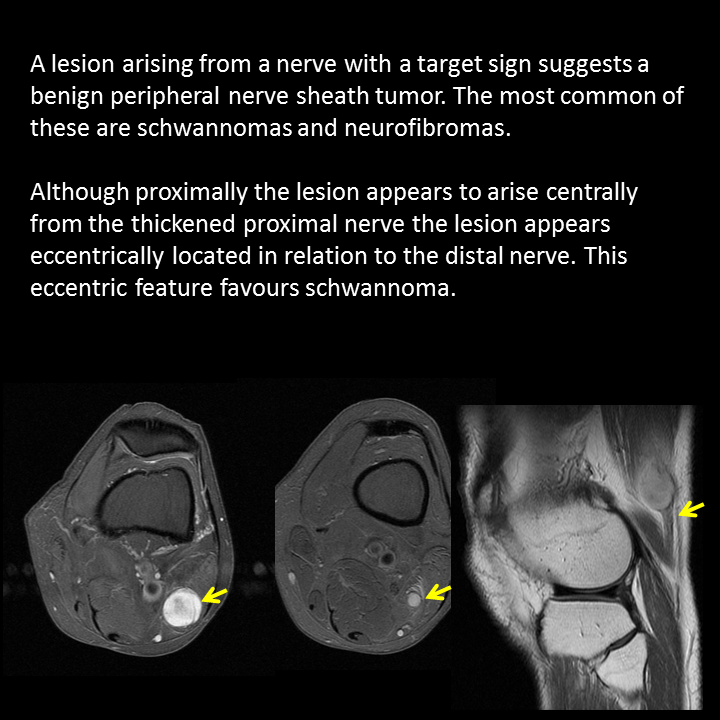
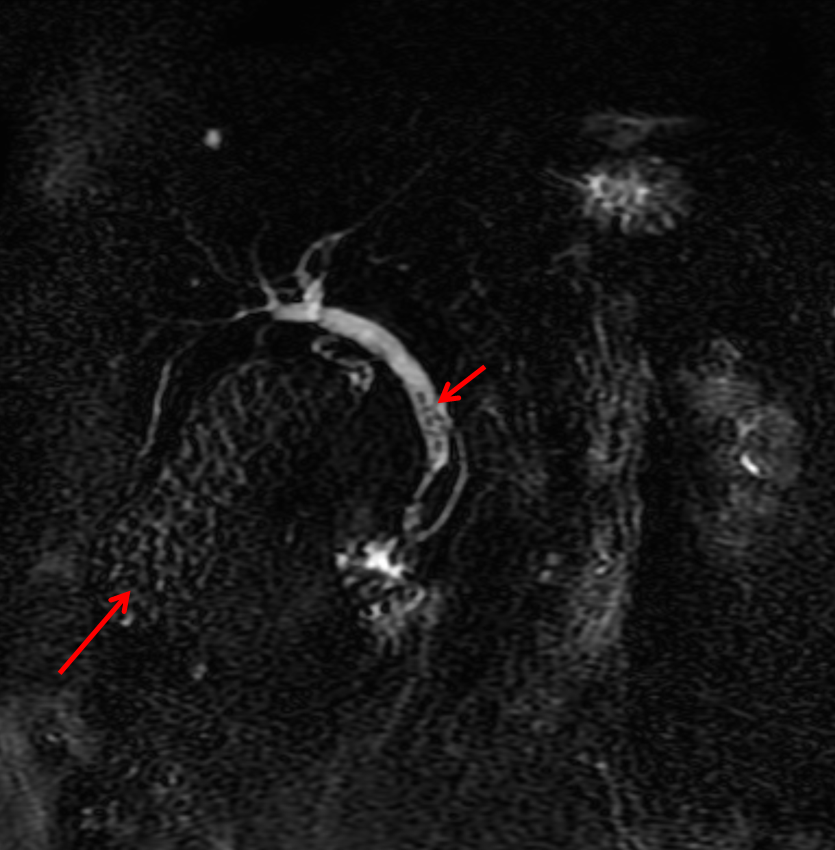
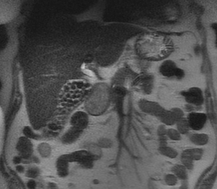
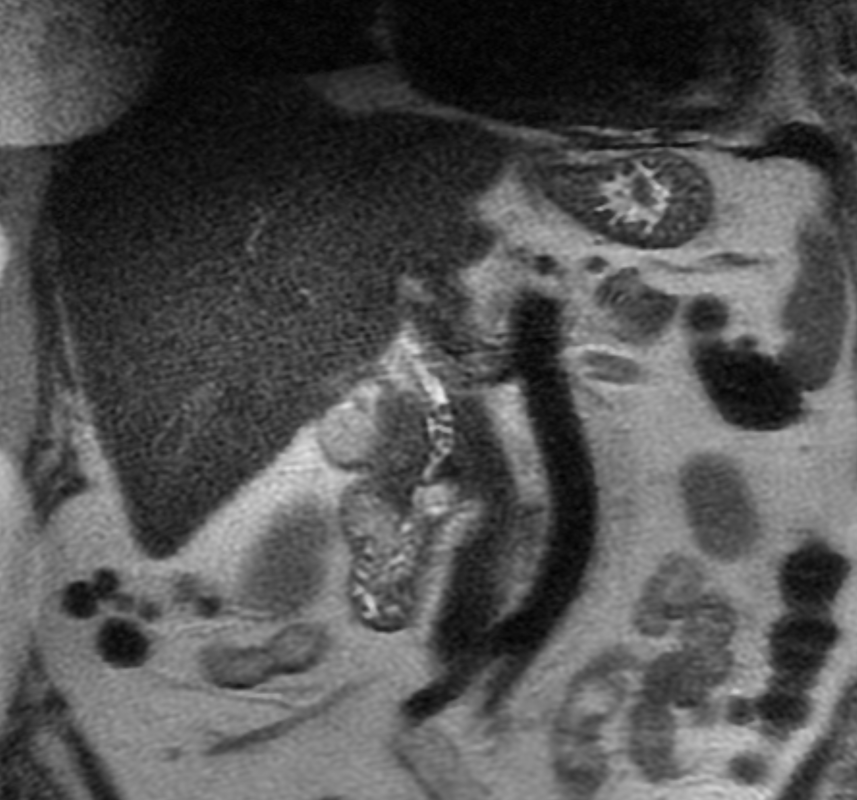
A much trickier case! The images provided from the initial Liver/MRCP examination are a T1 fat suppressed non-contrast image (top left), a FIESTA image (T2 equivalent bright blood sequence, top right), and two post contrast T1 fat suppressed images following intravenous gadolinium (second row). Thin and thick slab heavily T2 weighted MRCP images are provided on the third row. The final row demonstrates a post contrast T1 weighted fat suppressed image at 3 months. The FIESTA image has some degradation due to artefact from the lung parenchymal air affecting the right liver lobe. The original imaging demonstrates a left liver lobe T1 hypointense and heterogenously T2 hyperintense lesion. The lesion demonstrates both peripheral enhancement but also internal extensive reticular enhancement. In addition the contrast enhanced images demonstrate that there is a tubular low T1 abnormality with peripheral enhancement in the central right liver lobe. The MRCP images demonstrate that there are normal appearances of the gallbladder, common bile duct and pancreatic duct but that the intrahepatic ducts demonstrate small clusters of focal subsegmental duct dilatation. At 3 months, without any reported therapy the lesion has regressed to a smaller nodule of enhancement with some retraction of the liver capsule. The tubular low T1 abnormality and the associated enhancement are less conspicuous. The appearances of the tubular abnormality in conjunction with the MRCP images indicate intrahepatic duct dilatation. However, the intrahepatic duct dilatation is discontinuous and not secondary to any obstructive stones or stricture. One could consider the possibility of primary sclerosing cholangitis, however, no typical beading is demonstrated. Additionally the dilatation demonstrates enhancement. This is a feature that indicates inflammation or possibly infection. What about the enhancing lesion? Well the appearance of this abnormality could indicate a malignant lesion. One could then consider perhaps a case of cholangiocarcinoma with metastases, but again the non-contiguous nature of duct dilatation would not make sense and with regards to the lesion itself it is difficult to believe a malignant lesion regressed with no specific treatment. These appearances and the regression are typical of infectious processes, in particular recurrent pyogenic cholangitis. The focal lesion is termed an inflammatory pseudotumour and often demonstrates this avid and reticular enhancement. Regression can be spontaneous or in response to antibiotics and recurrence of lesions is common. Healing can result in focal scarring as in this case. Recurrent pyogenic cholangitis was previously referred to as oriental cholangiohepatitis and clinically is often associated with recurrent pyogenic episodes, although may be asymptomatic in the elderly. Intrahepatic marked ductal dilatation with intrahepatic duct stones can develop (hepatolithiasis). The stones are typically calcium bilirubinate. Infection of the bile with E Coli, Clonorchis sinensis or Ascaris Lumbricoides has been postulated as a cause. The condition remains more common in the far east although is not limited to that population. Long term complications include cholangiocarcinoma and intraductal papilloma formation. Radiology Level: FRCR, FRCS, MRCP, ABR, EDiR, Radiology Junior + The initial images are from an MRCP (magnetic resonance cholangiopancreatography) examination. The images on the left are T2 axial images, the images on the right from a FIESTA sequence (equivalent to FISP/TRUEFISP sequence, T2 predominant-although actually ratio of T2/T1- fat suppressed, bright blood sequence) demonstrating that the gallbladder is completely packed with low signal abnormalities consistent with stones. There is no apparent gallbladder wall thickening, oedema or pericholecystic inflammation. These appearances indicate chronic gallstone disease.
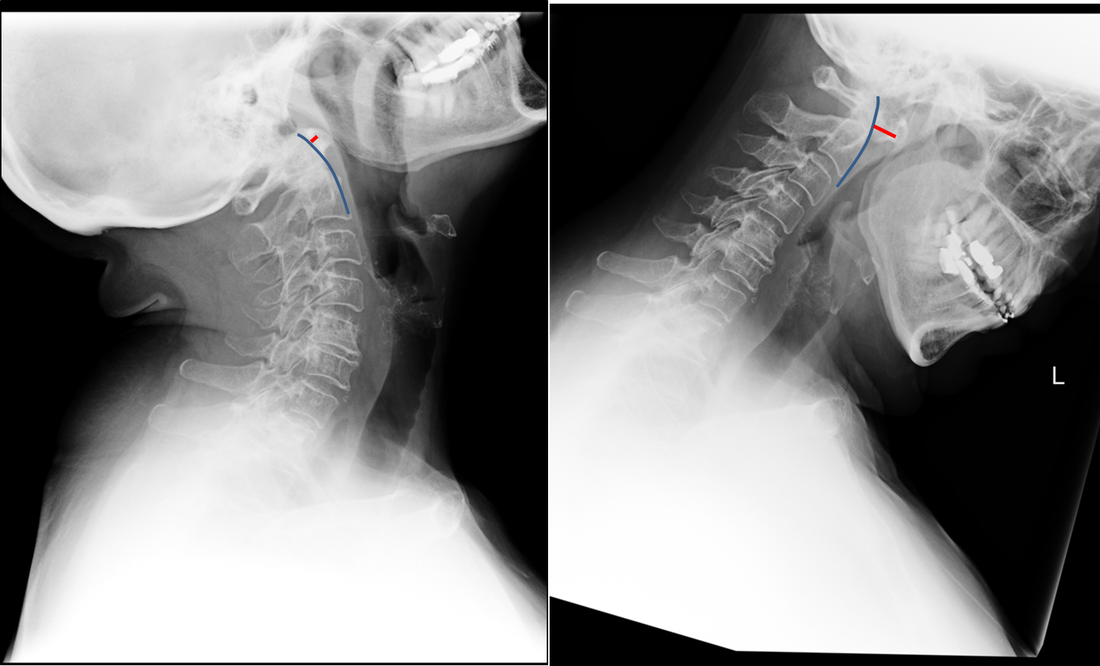
It is important, however, to carefully review the remainder of the biliary tree. Hopefully, on such a review you would have also assessed the common bile duct which can be seen in the head of the pancreas (short narrow). This is mildly dilated but additionally on both the images dependent low signal abnormality can be identified consistent with bile duct stones i.e. choledocholithiasis. The presence of gallstones within the bile duct is an important parameter to indicate prior to laparoscopic surgery to ensure that the duct is cleared. The appearances are very evident on the oblique T2-weighted radial ray projection (left). In this image it is perhaps easy to not see the gallbladder due to the loss of high T2 signal caused by multiple gallstones (long narrow). In this instance the choledocholithiasis is easily appreciated (short narrow). These are also appreciated on coronal SSFSE (single shot fast spin echo) acquired through the gallbladder and the lower biliary tree. In identifying extrahepatic duct choledocholithiasis, particularly single stones, it is important to exclude artefact related to flow. Flow artefacts do appear central and are not consistent whereas choledocholithiasis is typically dependent and reproducible on more than one sequence. A typical site for flow artefacts is also at the crossover of the right hepatic artery over the common hepatic duct near the hepatic duct confluence. Radiology Level: FRCR, FRCS, MRCP, ABR, EDiR, Radiology Junior + The images demonstrate a pair of cervical spine radiographs. The image on the left is in extension the image on the right in flexion. Both images are adequate visualising to C7-T1 or lower. The extension image demonstrates that there is some moderate degenerative change with sclerosis and joint space reduction at C5-C6. The flexion film demonstrates that there is straightening of the cervical spine at this level in flexion. There is, however, no spondylolisthesis (slip).
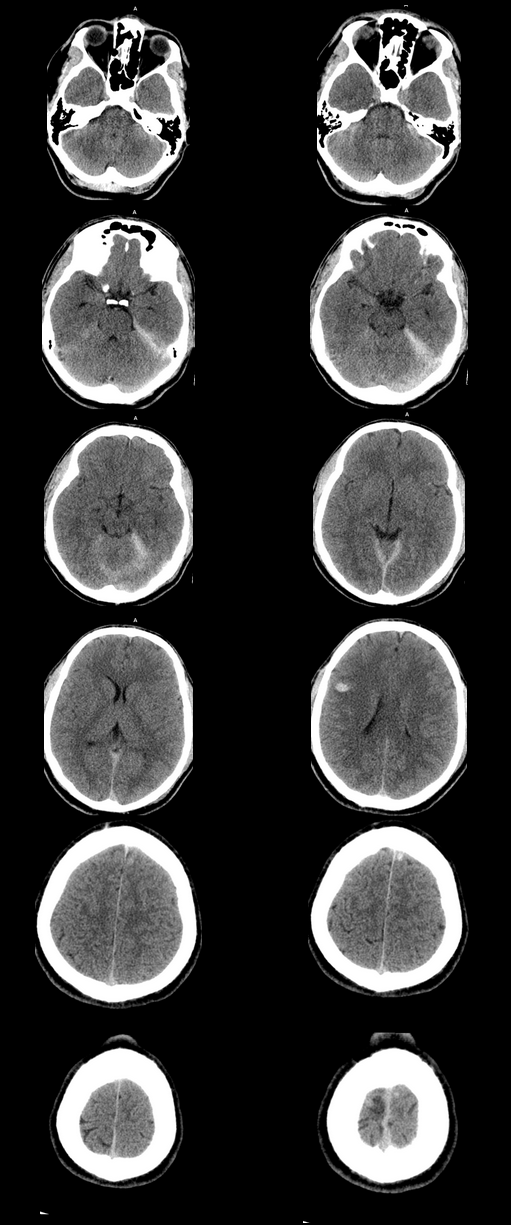
Looking more carefully at the atlanto (C1) –axial (C2) articulation, we can see that in extension the anterior margin of the dens can be traced superiorly to the skull base (blue line). The predental space (also called atlantodental or predentate space) measures only 2mm (red line). In flexion though the predental space greatly increases to >7mm. The appearances are those of non-traumatic atlanto-axial subluxation/dislocation. In addition there is some loss of clarity of the odontoid peg (confirmed on MRI/CT) suggesting some erosion. These appearances were due to rheumatoid disease. The patient was being assessed pre-operatively. Typically the predental space is usually <=3mm in neutral or extension radiographs, <=5mm in children. In children the ligamentous laxity of the neck in flexion allows some minor increase of this space, but not in adults. The anterior motion of the atlas on the axis/dens is typically restricted by the transverse ligament. The disruption of this ligament can occur by trauma (look for associated fractures or prevertebral soft tissue swelling) or by inflammatory conditions (typically rheumatoid disease but also less commonly psoriasis, Down’s and Morquio’s syndromes, Neurofibromatosis, Osteogenesis Imperfecta). Disruption of the transverse ligament enables atlanto-axial instability. This can be asymptomatic or life-threatening (particularly in trauma). Specifically, posterior translation of the dens can compress the cord resulting in upper motor neuron compressive neurology. Such unexpected spinal compression may become first apparent during intubation and is, therefore, screened for in susceptible patients with rheumatoid arthritis. The condition is termed atlanto-axial instability/subluxation or dislocation and should be differentiated from atlanto-axial rotatory subluxation in which the primary injury is a rotation at C1-C2 that may be associated with unilateral facet dislocation, fractures and nerve root compression with flexed rotated angulated neck malalignment. Case to Ponder 79 Answer: Acute subdural, petechial haemorrhages and early diffuse axonal injury29/1/2017 Radiology Level: FRCR, FRCS, MRCP, ABR, EDiR, Radiology Mid-Level ++ The appearances here are of a severe intracranial injury.
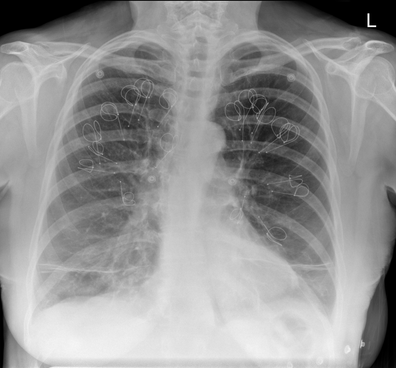
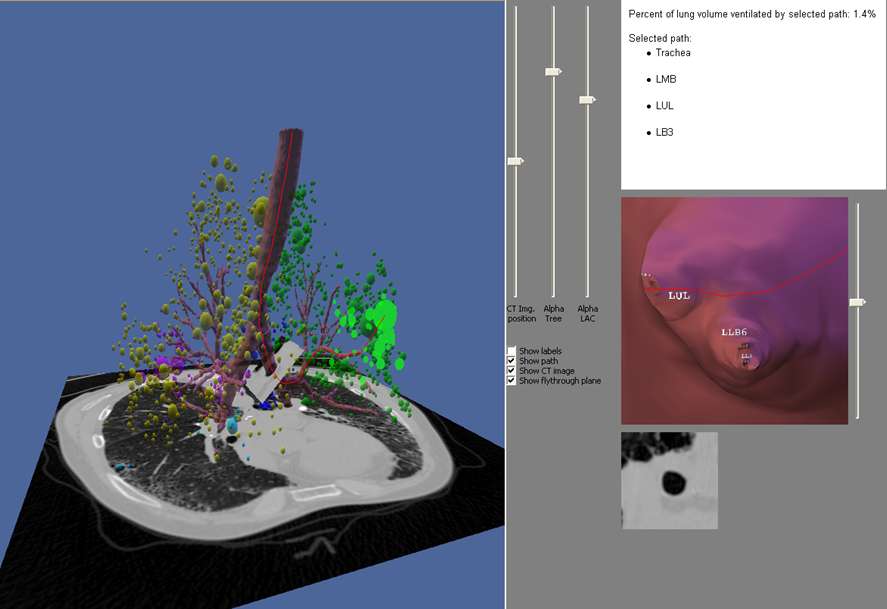
Radiology Level: FRCR, ABR, EDiR, MRCP, Radiology Senior +++ The appearances demonstrate multiple metallic loop structures in the upper lobes with extensive bibasilar atelectasis. These findings are due to endobronchial bronchoscopic insertion of nitinol endobronchial coils for the minimally invasive treatment of emphysema. The principal of usage of these colis is that by reducing air flow to emphysematous diseased segments of lung, that there will be improved air fluid to more functional lung parenchyma and overall this will result in improved gas exchange and hence lung function. The procedure is intended to improve lung function patients with upper or lower lobe heterogeneous emphysema. These endobronchial coils are inserted into the airways by bronchoscopy under sedation or general anaesthesia. A guide wire is inserted through the bronchoscope under fluoroscopic guidance. A catheter is passed over the guide wire and the straightened coil is introduced through the catheter with the catheter withdrawn as the coil held in place using grasper. When released the straightened coil reverts to to predetermined shape pulling the surrounding disease tissue and reducing lung volume. Usually up 5-15 coils are inserted per treated lobe (usually the upper lobes). Each lung is treated separately as there is a risk of pneumothorax. The coils remain permanently in location. This type of volume reduction surgery is an alternative to patients undergoing formal lung volume reduction surgery or endobronchial valve insertion. The validity of such usage has been supported by small trials that suggest principally improved symptomatology, rather than measurable improved pulmonary function, after insertion versus control patients. The usage has been supported in the UK by NICE guidance. My entirely anecdotal experience, supported by radiological imaging is that this is entirely bogus! Emphysema at CT is either focal, or diffuse and heterogeneous. If it is focal and compressing normal lung, lung volume reduction surgery makes sense and I have seen real improvement. Endobronchial valves should theoretically work but tend in my experience to become obstructed by lung becoming atelectatic adjacent to them. Both these procedures can be supported by lung CT that specifically guides by virtual bronchoscopy to the areas to resect, or the particular airways to obstruct or bypass by a valve (see image below). In contradistinction these valves may I am sure help the occasional patient who has diffuse emphysema in the upper lobes, by reducing ventilation and perfusion to these areas. But for most people with "heterogeneous emphysema" on CT have a bulla here and a bulla there throghout the lungs. Randomly inserting some coils is just as likely to reduce ventilation and perfusion to a good subsegment of lung as to an emphysematous portion.
It, therefore, comes to me as no surprise that a recent large study in JAMA casts doubt even on the symptom based improvement of these devices (6 minute walks, questionairres), suggesting much of this must be placebo related. Importantly it states that the treated groups unsurprsingly suffer considerably more pneuthoraces and infective exacerbations. Regardless these will be around for a while and need to be recognised. This patient suffered from recurrent haemoptysis, likely due the coils. Reference: Sciurba FC et al. JAMA. 2016 May 24-31;315(20):2178-89. doi: 10.1001/jama.2016.6261. Effect of Endobronchial Coils vs Usual Care on Exercise Tolerance in Patients With Severe Emphysema: The RENEW Randomized Clinical Trial. https://www.ncbi.nlm.nih.gov/pubmed/27179849 Radiology Level: FRCR, ABR, EDiR, FRCS, Radiology Senior +++
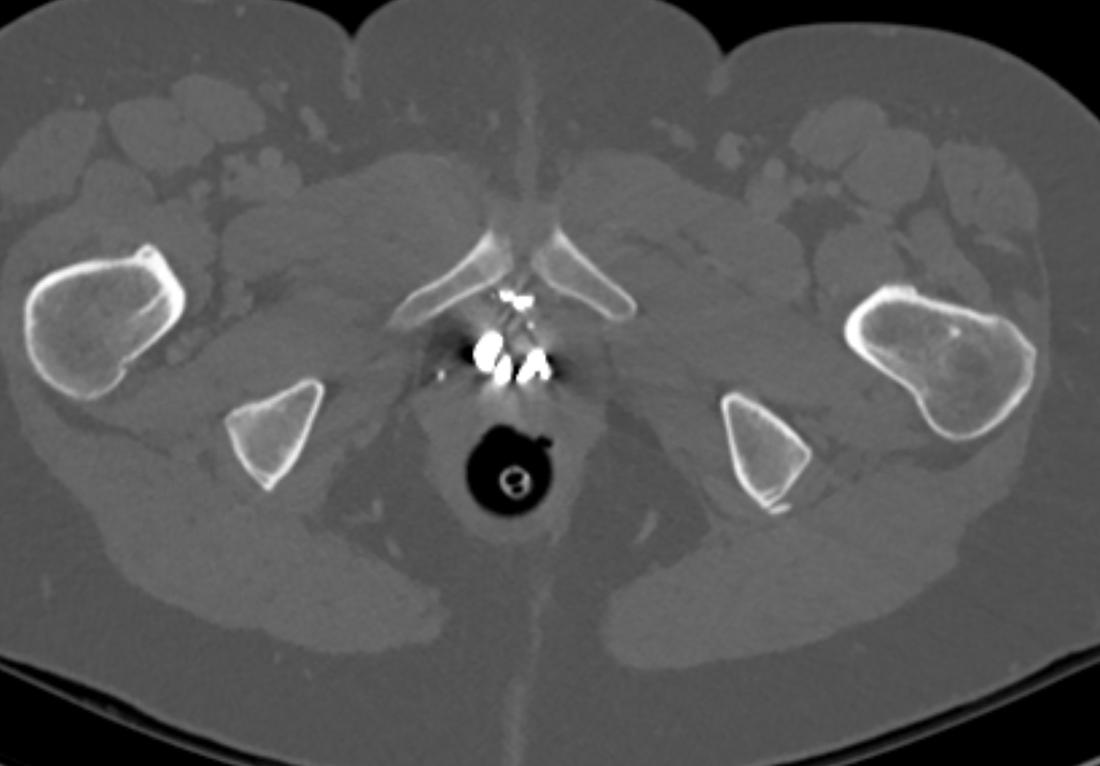
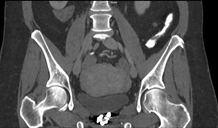
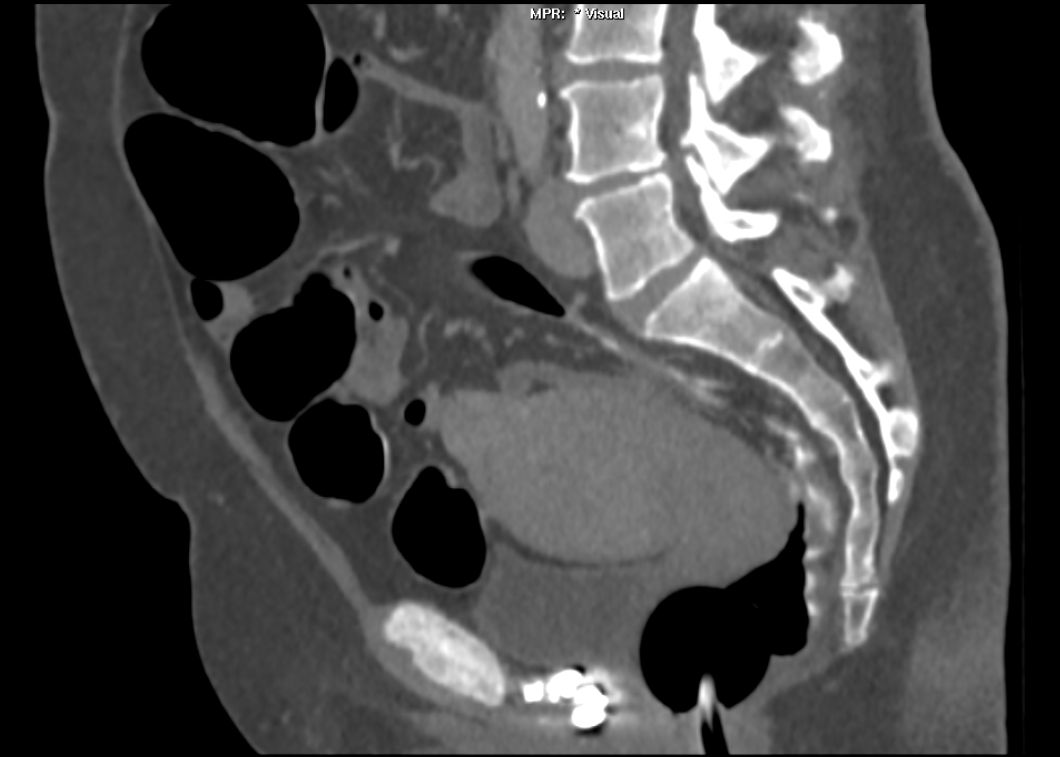
The images demonstrate high density clustered foci within the central lower pelvis. A few further foci noted in isolation along the pelvic sidewall. These appearances are too low central to reflect typical ureteral calculi and too inferior to reflect typical prostatic or seminal vesicle calcifications. The bladder outlines can be identified full of urine and the high density foci are noted to be below this, suggesting these are not bladder calculi. Indeed the density of these abnormalities far exceeds that of bone suggesting that these are metallic or other similar high density. The anatomic location is inferred from the abdominal radiograph but can be further delineated from the additional CT images provided. These demonstrate the presence of high density material below the bladder, surrounding the bladder neck. These appearances are due to periurethral injection of bulking agents in this case Duraspeheres. These agents are used to tackle stress urinary incontinence in female patients. In this instance the abnormality is due to a pyrolytic coated beads which stimulate collagen formation and improved urinary continence. The additional occasional sidewall high density focus is due to vascular extravasatiion which should be avoided. Bulking materials previously used included autologous fat. However, this has resulted in anecdotal fatal lipid pulmonary embolism and is no longer advised. Injections are targeted into the submucosal tissues of the urethra and bladder neck under anaesthesia. Radiology Level: FRCR, ABR, EDiR, MRCP, Radiology Senior +++
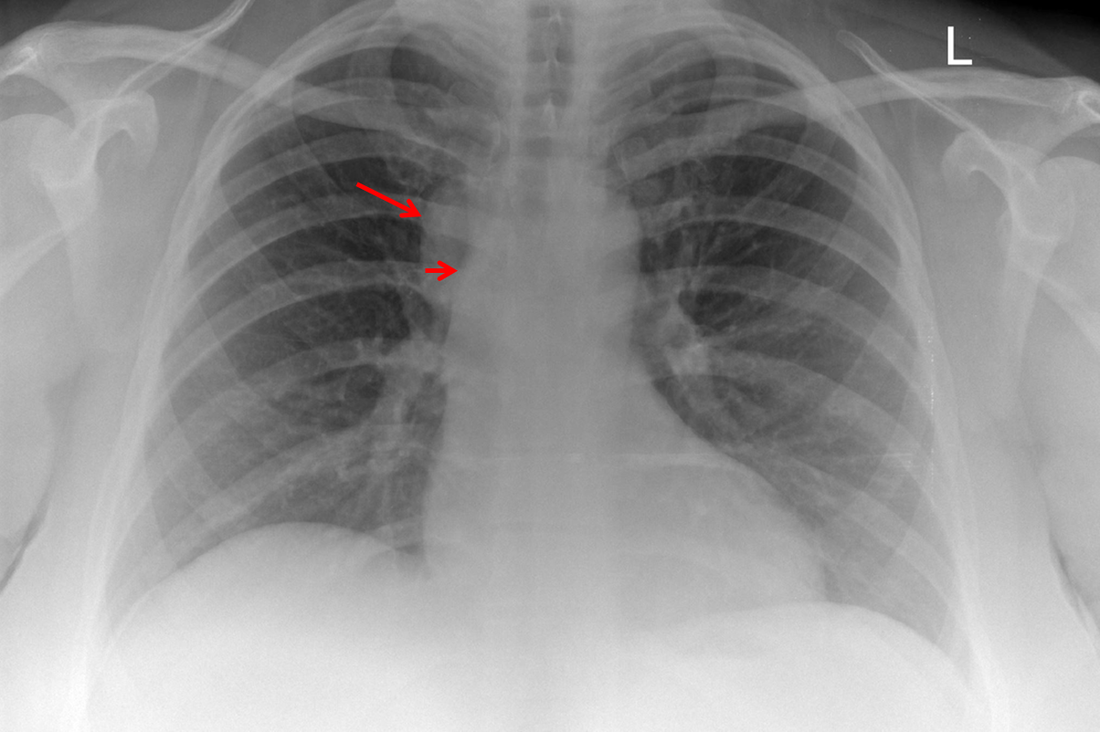
The initial chest x-ray demonstrates a right paramediastinal opacity confluent with and widening the right paratracheal stripe. Compare this case to case 75. In that case the right paramediastinal mass opacity did not efface and widen the right paratracheal stripe and so was not anatomically right paratracheal. The current case chest radiographic appearances suggest that the findings are due to a mass lesion adjacent to the right margin of the trachea. Is the rest of the chest x-ray normal? Well actually no it is not. Look at the more superior trachea. It is displaced to the right, suggesting a more superior cervical. The most common cause for this would be a thyroid goitre. Could this be causing both the left and right paratracheal abnormalities? Now let us review the CT imaging. We can see that there is enlargement of the left lobe of the thyroid and that accounts for the displacement of the trachea to the right. As suggested on the chest x-ray there is indeed a right paratracheal abnormality and this is confirmed on the coronal reconstructions explaining why the chest radiograph right paratracheal stripe is widened. In viewing the characteristics of the right paratracheal tissue it is clear that this has a very similar consistency to the left paratracheal thyroidal tissue, containing heterogenous low attenuation areas as well as a focal calcification. However, axial and coronal images demonstrate that there is no connection between the inferior margin of the right thyroid and the right paratracheal mass. What is the differential for these appearances? Well we can consider causes of anterior mediastinal masses. However, the appearances are too heterogenous for a diagnosis of lymphoma. Too lateral for a diagnosis of thymoma or a teratoma. In this location we should also consider a focal enlarged node, due to metastatic disease or perhaps due to granulomatous aetiology. However, no other nodes were present elsewhere. Other rarer lesions to consider would include ectopic parathyroid lesions or paraganglioma lesions, although both these are more typically hyperenhancing. So what does this leave is with? Well the lesion looks exactly like a thyroid goitre even though it is not connected to the right lobe. Well if it looks like a duck and quacks like a duck the saying is it usually is a duck! My thought was that this was ectopic thyroid with a goitre. The lesion processed to biopsy by endobronchial ultrasound for confirmation revealing typical colloid material suggestive of a thyroid goitre. The biopsy confirms a benign goitre in ectopic mediastinal thyroid tissue. Ectopic thyroid tissue is not uncommon, occurring during in the embryonal migration of thyroid tissue from the base of the foramen caecum to the typical pretracheal region. However inferior mediastinal ectopy is far more uncommon, likely less than 1% of ectopic cases, although the right paratracheal location has been reported. This should be differentiated from a multinodular thyroid gland with a small atretic band from the right lobe to the right paratracheal mass. Typical thyroid goitres extending from the neck descend in the right paratracheal or left paratracheal location. It is usually only the rarer isthmic lesions that extend anteriorly into the anterior mediastinum as the list books suggest. This case was primarily included as a companion training exercise for case 75 to evaluate the right paratracheal region on chest radiographs. Compare these chest radiographs, the corresponding coronal CTs, to improve your interpretation of the right paratracheal region. Radiology Level: FRCR, ABR, EDiR, MRCP, Radiology mid-level ++ The original chest x-ray demonstrates a large hemispherical opacity lateral to the right superior mediastinal border with a well defined lateral margin (long narrow). Assuming that most lesions are roughly spherical, it is therefore most likely to reflect a mediastinal lesion. It is important to recognise that there is no widening of the right paratracheal stripe. This right paratracheal stripe can still be observed extending along the inferior margin of the right tracheal border, extending to the tracheobronchial angle measuring no more than the upper limits of 5mm (short arrow). This implies that the lesion is not in the anterior mediastinum and must lie in the middle or posterior mediastinum. The for dislocation the finding is unlikely to reflect adenopathy or mass, for example due to lymphoma or other anterosuperior mediastinal mass.
The CT images confirm that there is a mediastinal abnormality that is well defined, homogeneous and of unform relatively low density, less than solid.The abnormality is unilocular and single. The abnormality lies in the middle/posterior mediastinum and as can be confirmed on the coronal reconstructions is separate from the trachea. Hence there is no silhouette sign, with no loss or widening of the right paratracheal stripe. These appearances are diagnostic of a bronchogenic cyst, also referred to as a foregut duplication cyst. These lesions are very common in this location and more typically right-sided, extending into the azygo-oesophageal region as in this case. Although the density of the abnormality is frequently fluid, the abnormalities may be slightly hyperdense, indicating a slightly proteinaceous contents. Similarly on MRI it is in my experience more typical to see mild T1 hyperintensity rather than the typical low T1 signal characteristics often described. No enhancement is noted within the abnormality on CT or MRI although occasionally mild enhancement of the wall of the cyst can be demonstrated. Most bronchogenic cysts are asymptomatic discoveries and often measure as much as 10 cm at presentation. Surgery is indicated for comparison symptomatology which is unusual. Secondary infection can be an occasional cause for presentation. The diagnosis should be suspected on plain films where there is a well-defined lesion in a roughly spherical configuration particularly in the middle/posterior mediastinum near the carina. |
From Grayscale
Latest news about Grayscale Courses, Cases to Ponder and other info Categories
All
Archives
October 2018
|

|
|
Grayscale Courses est. 2015


 RSS Feed
RSS Feed
