|
Radiology Level: FRCR, ABR, EDiR, FRCS, Radiology Senior +++
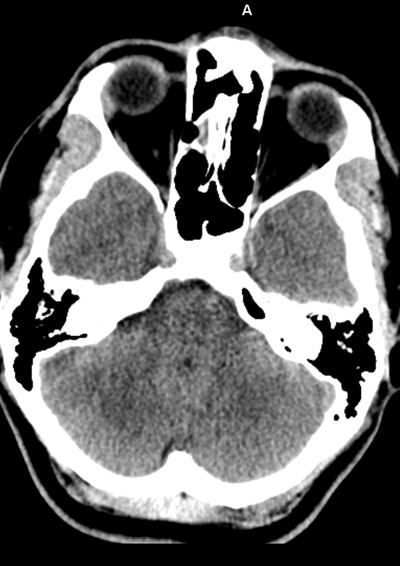
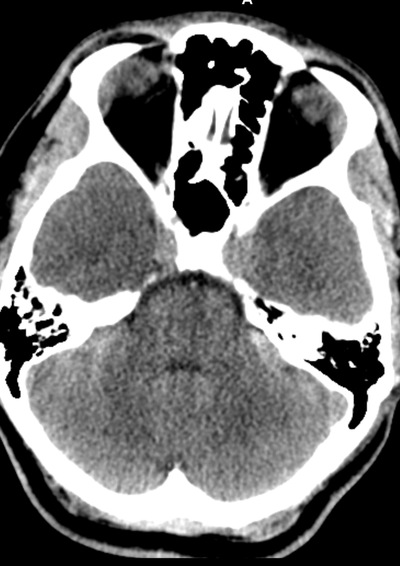
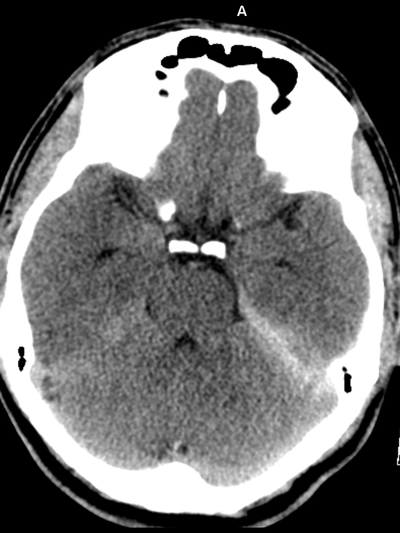
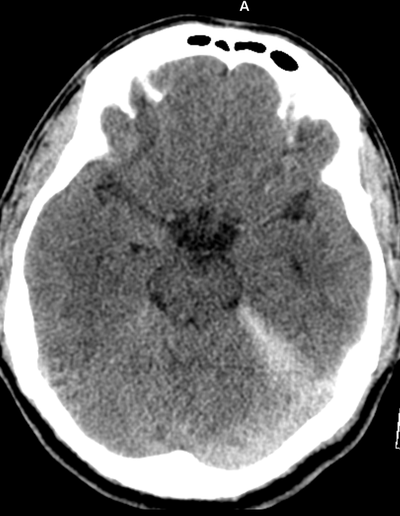
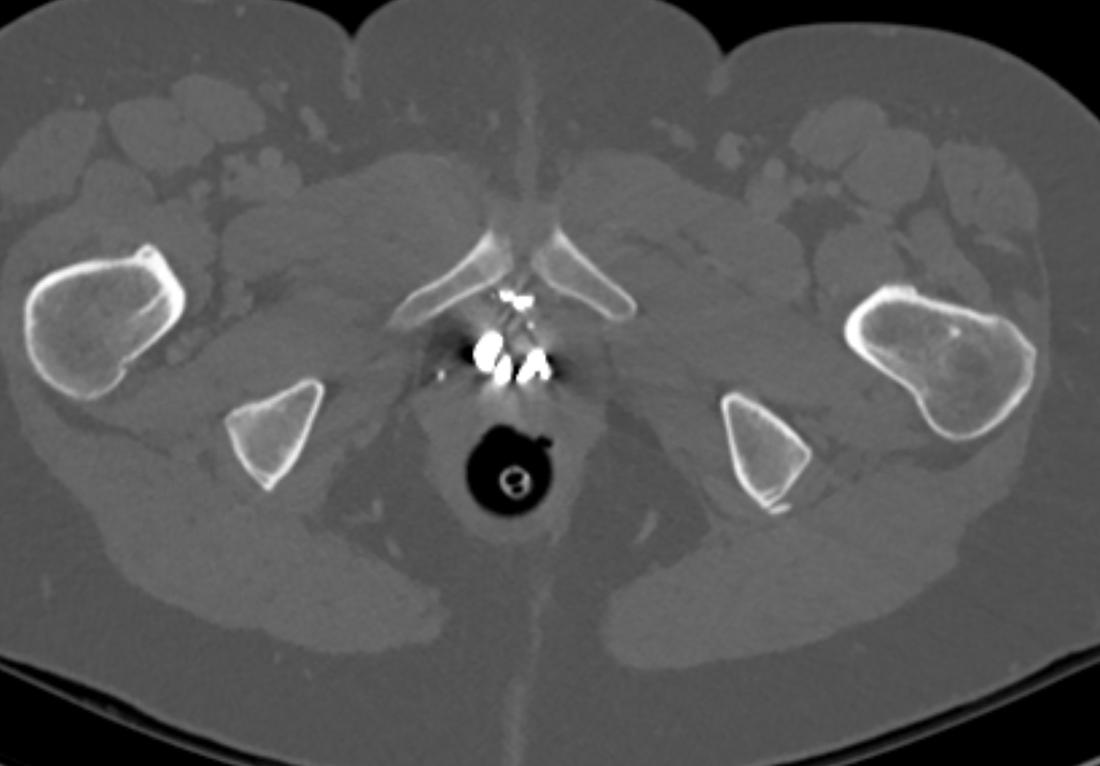
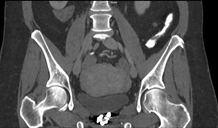
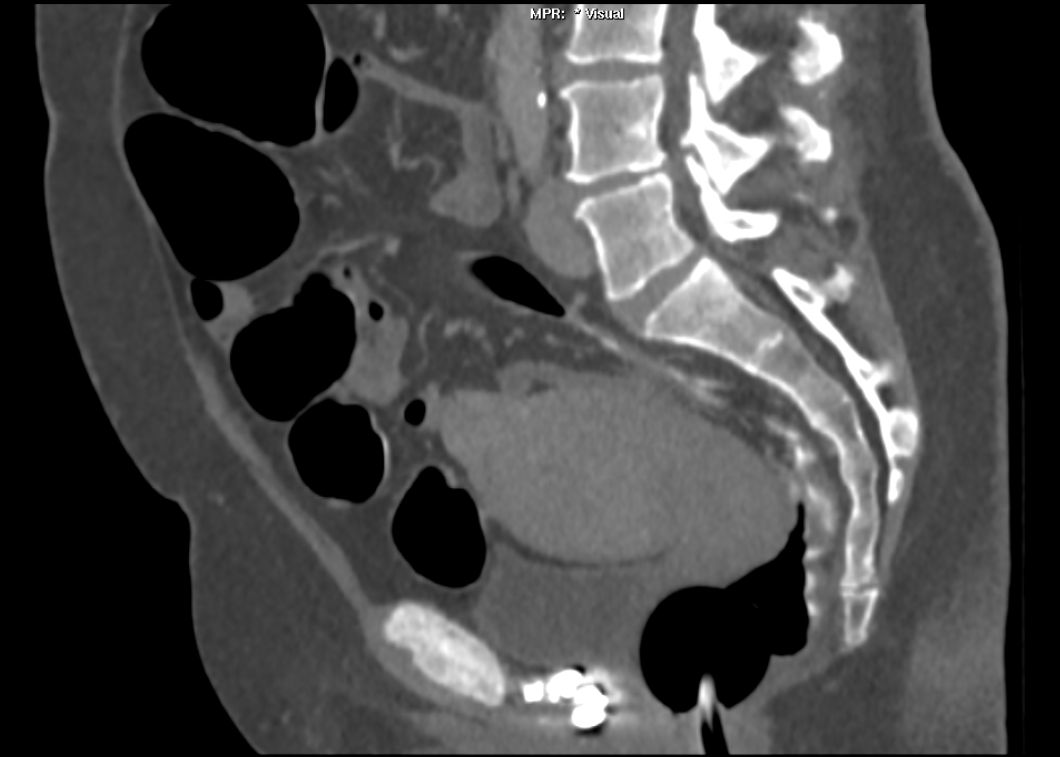
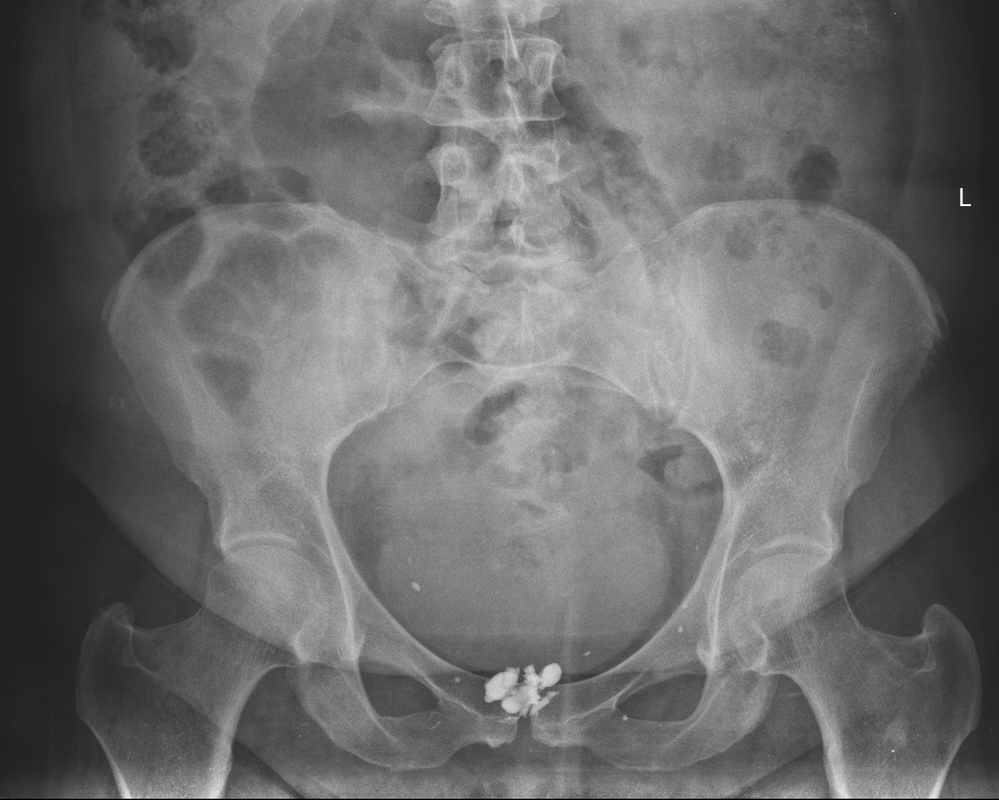
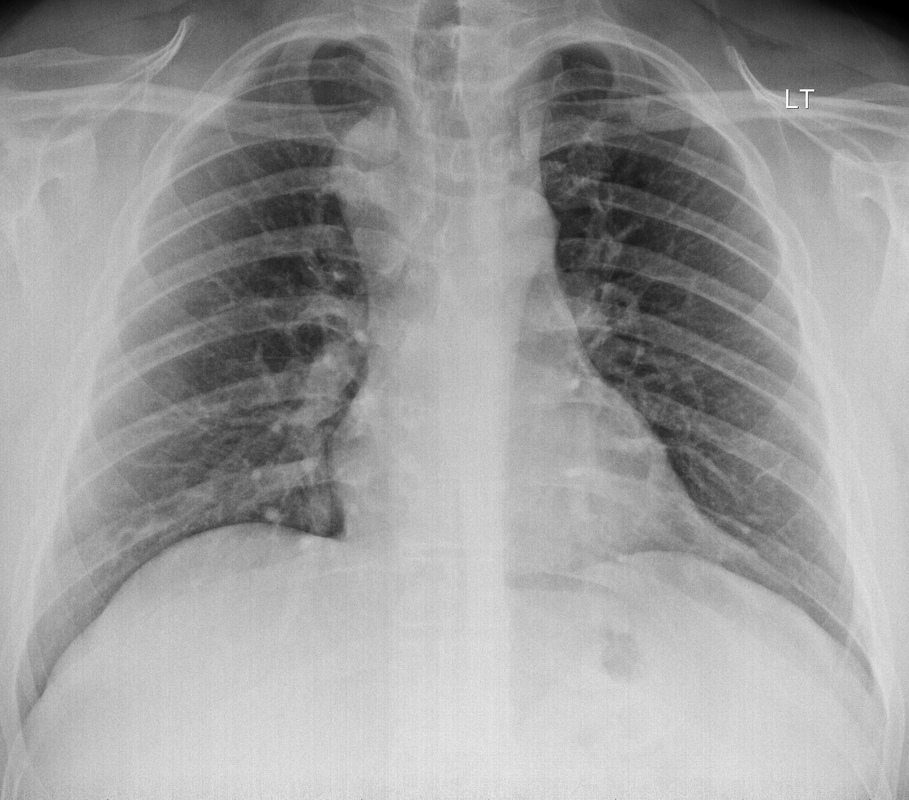
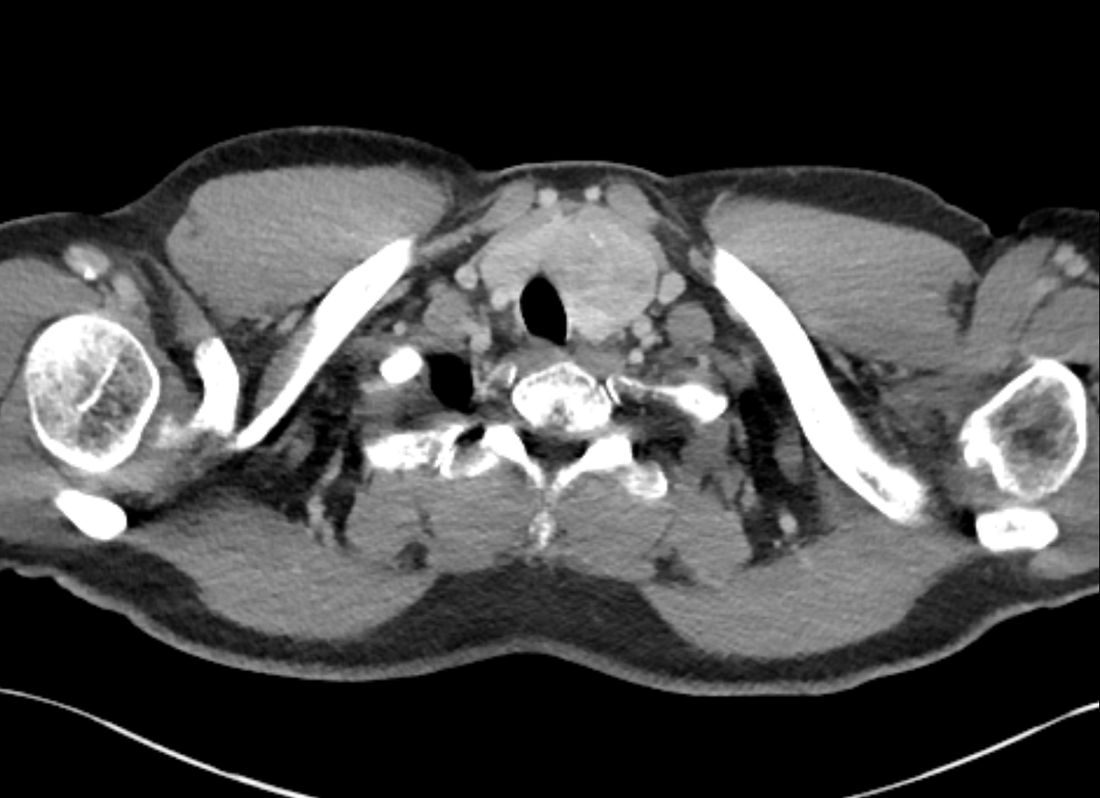
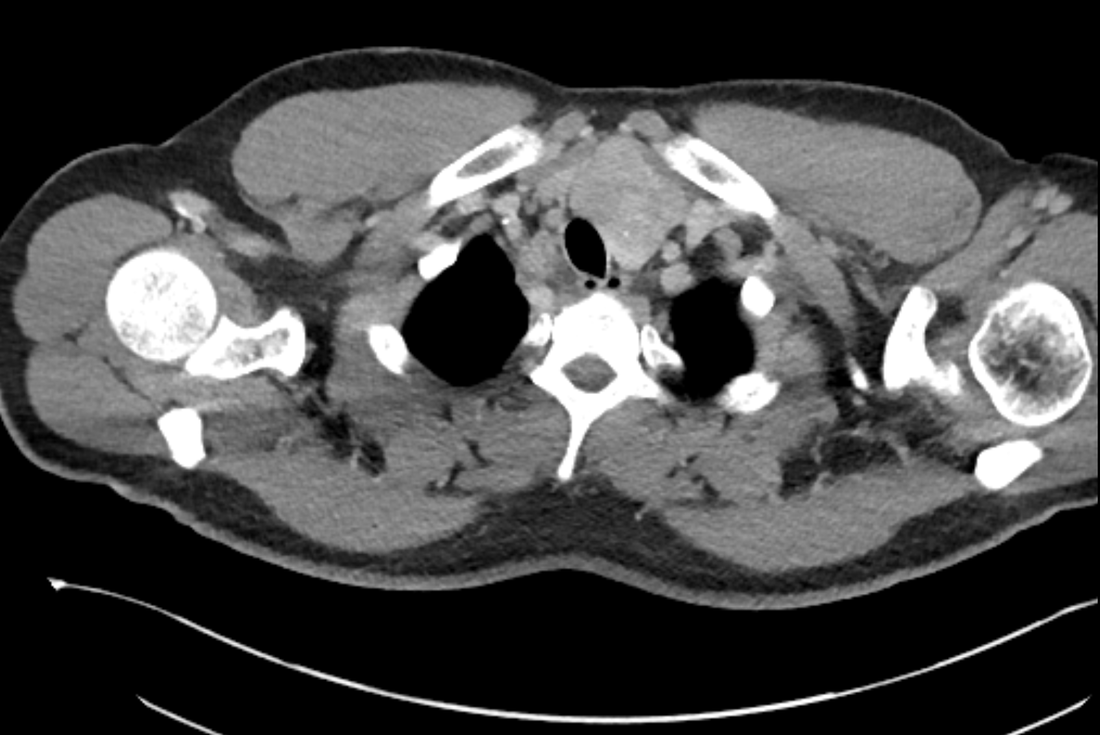
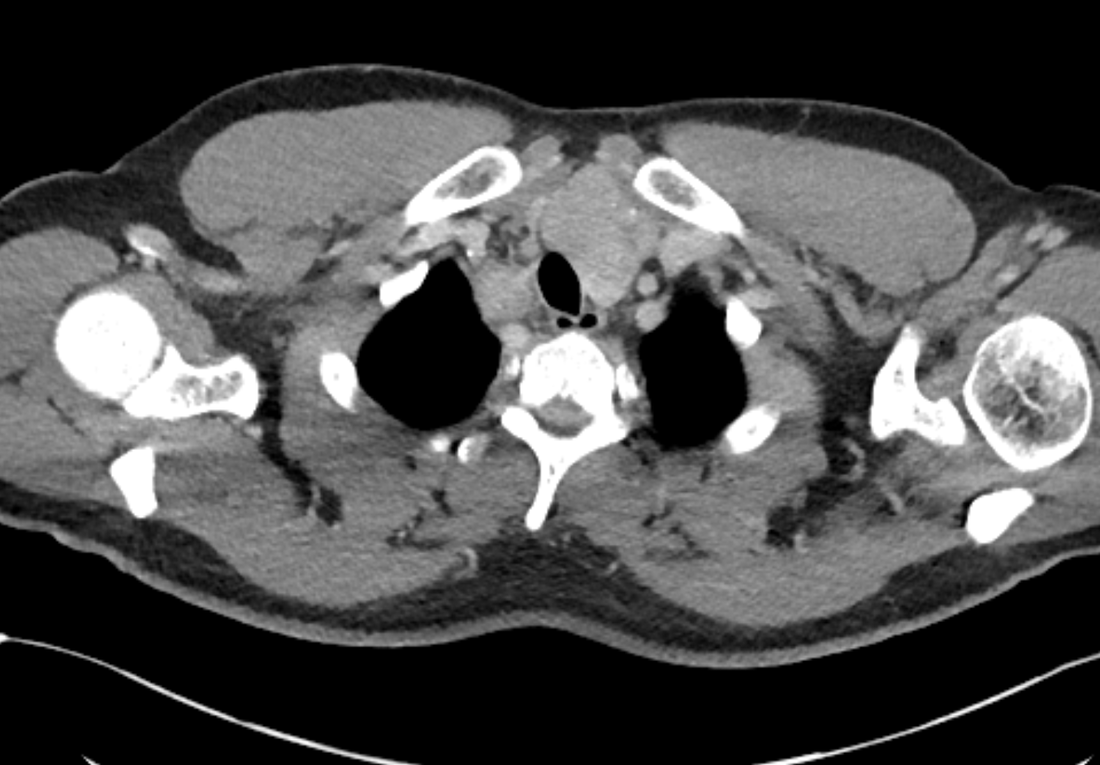
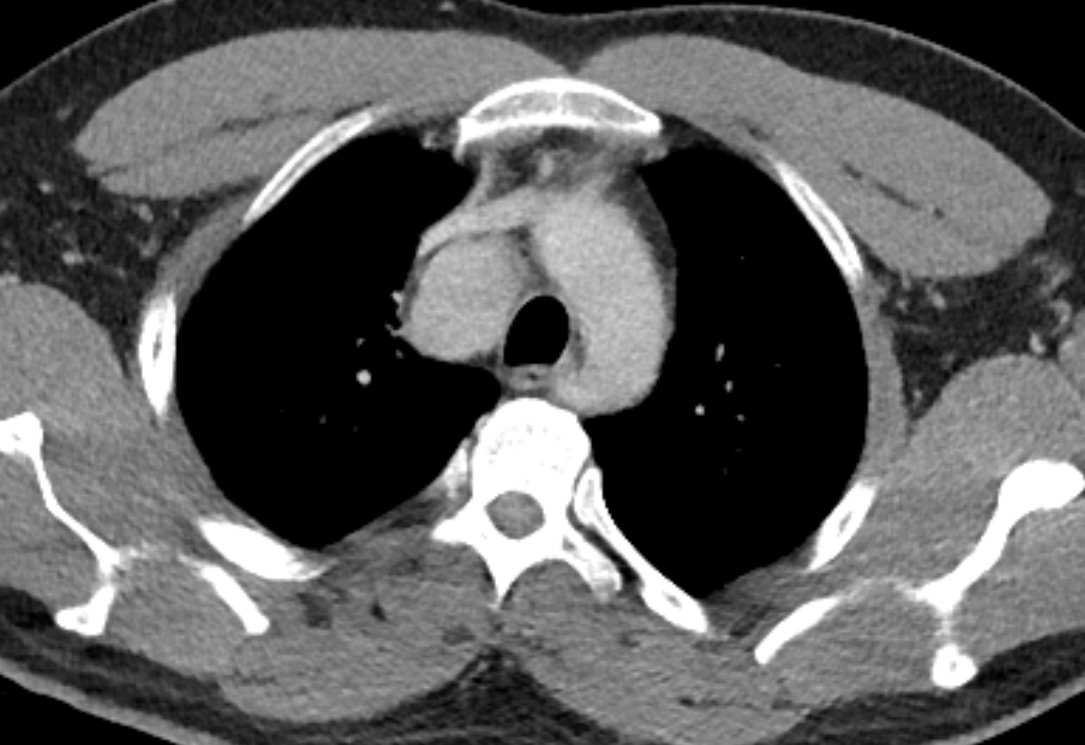
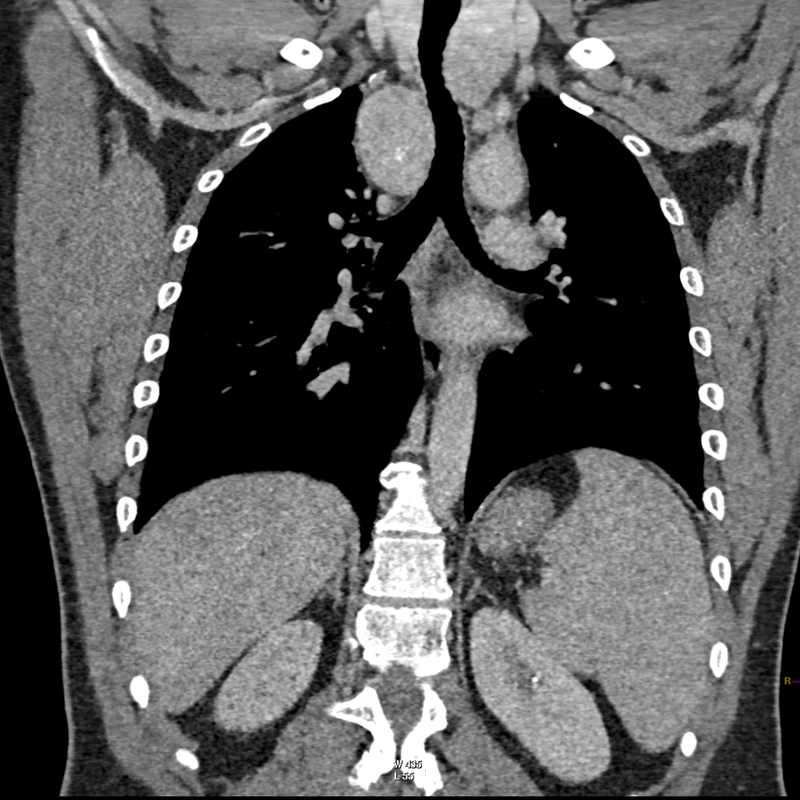
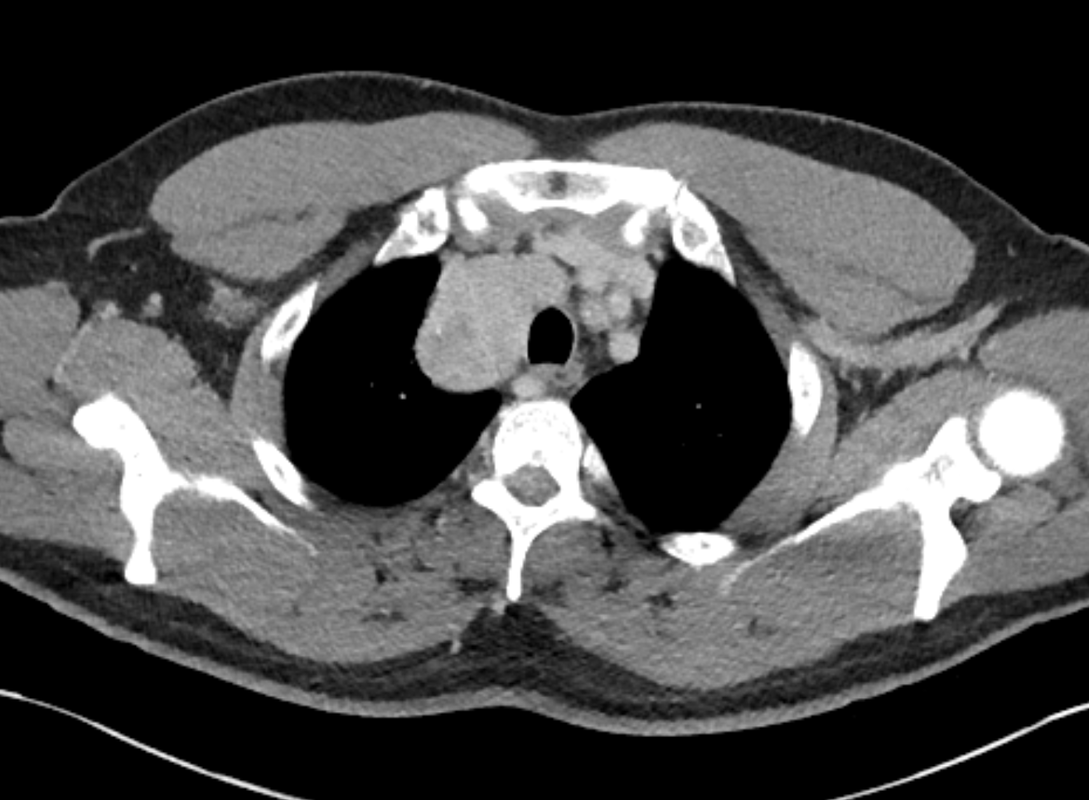
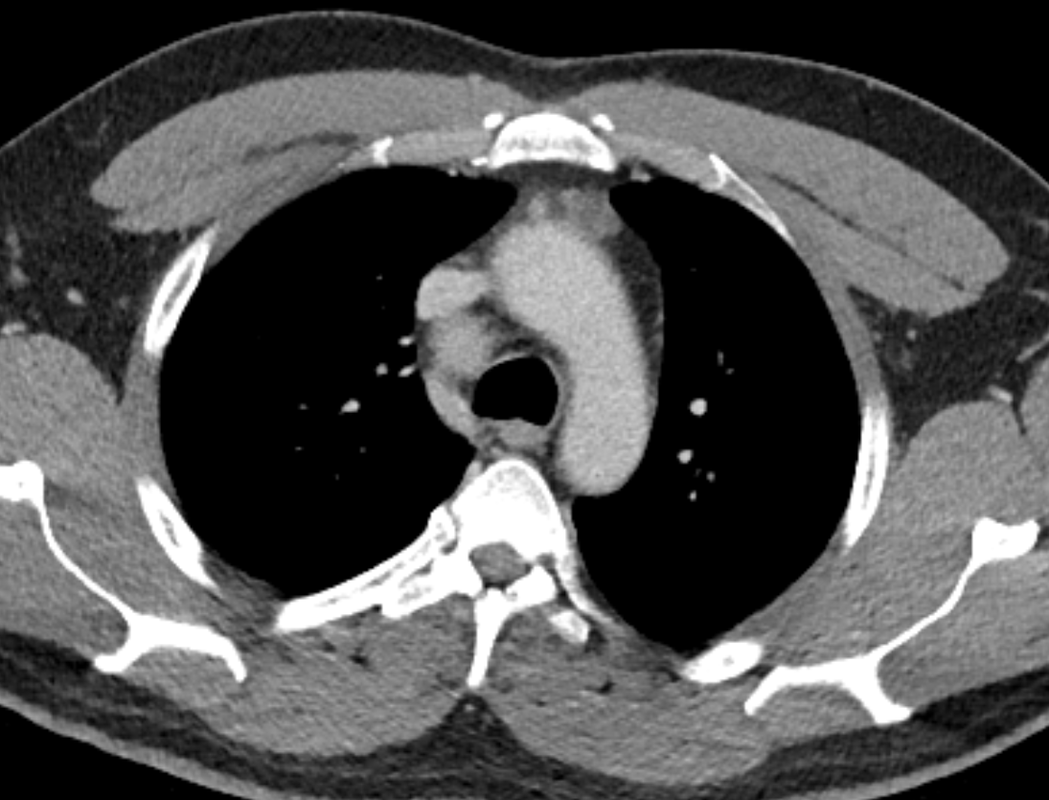
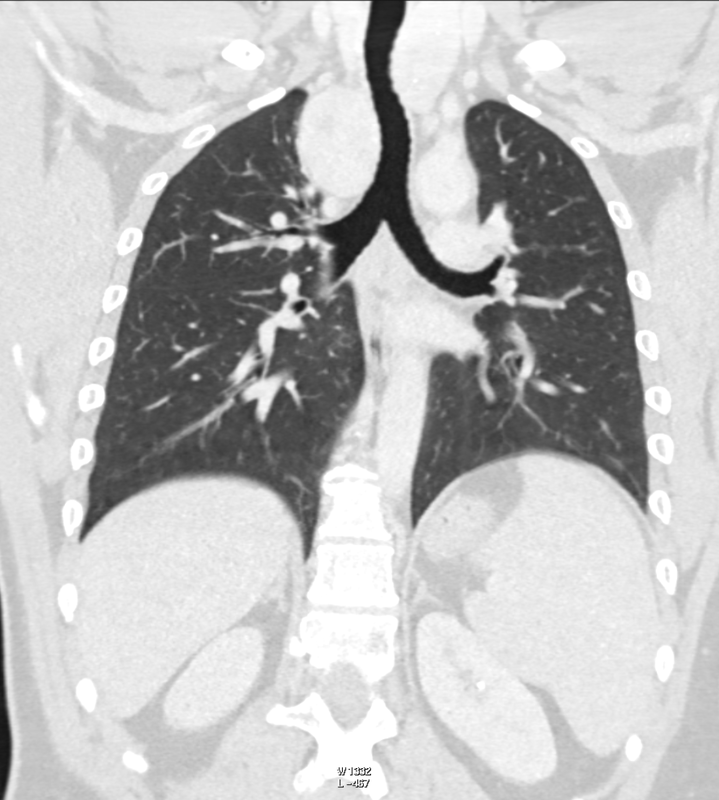
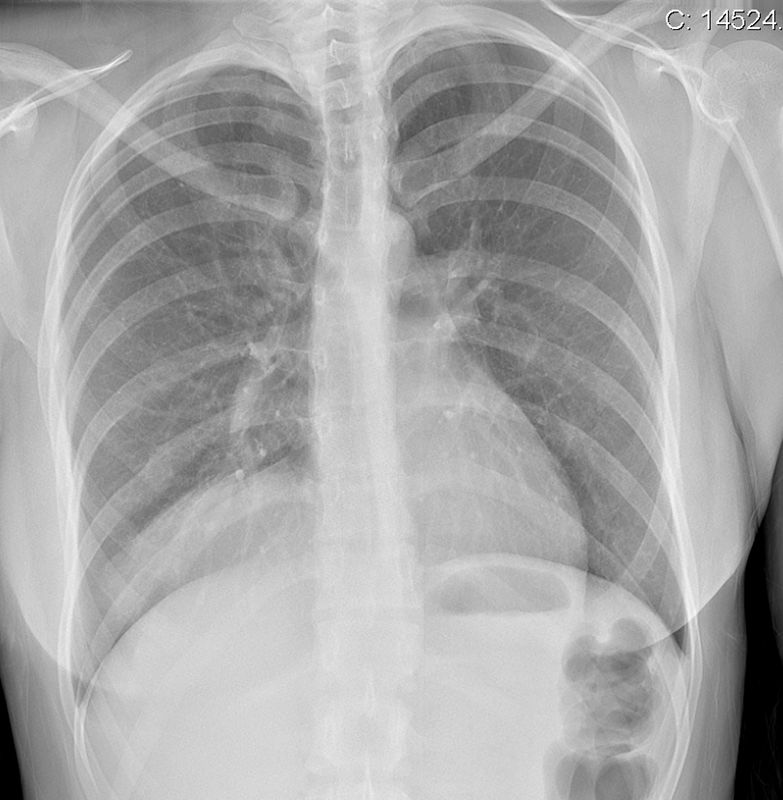
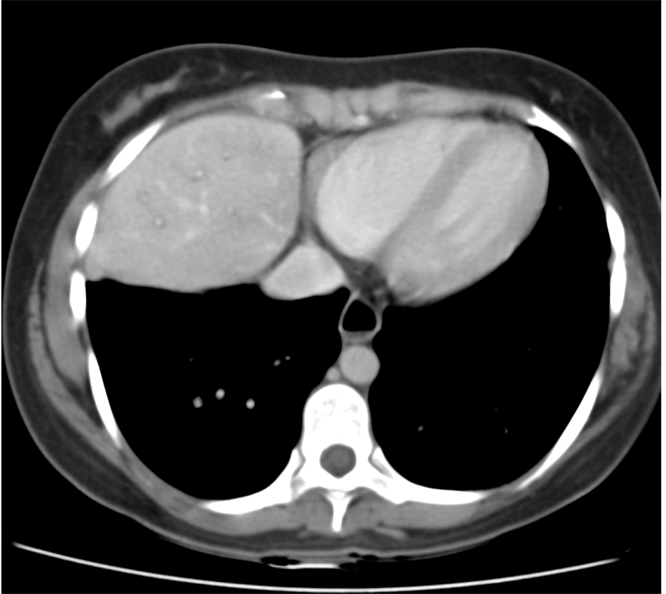
The images demonstrate high density clustered foci within the central lower pelvis. A few further foci noted in isolation along the pelvic sidewall. These appearances are too low central to reflect typical ureteral calculi and too inferior to reflect typical prostatic or seminal vesicle calcifications. The bladder outlines can be identified full of urine and the high density foci are noted to be below this, suggesting these are not bladder calculi. Indeed the density of these abnormalities far exceeds that of bone suggesting that these are metallic or other similar high density. The anatomic location is inferred from the abdominal radiograph but can be further delineated from the additional CT images provided. These demonstrate the presence of high density material below the bladder, surrounding the bladder neck. These appearances are due to periurethral injection of bulking agents in this case Duraspeheres. These agents are used to tackle stress urinary incontinence in female patients. In this instance the abnormality is due to a pyrolytic coated beads which stimulate collagen formation and improved urinary continence. The additional occasional sidewall high density focus is due to vascular extravasatiion which should be avoided. Bulking materials previously used included autologous fat. However, this has resulted in anecdotal fatal lipid pulmonary embolism and is no longer advised. Injections are targeted into the submucosal tissues of the urethra and bladder neck under anaesthesia. The initial chest radiograph demonstrates an abnormal opacity in the right inferior hemithorax. This demonstrates a parallel orientated opacity to the right hemidiaphragm, with some very medial loss of visualisation of the right hemidiaphragm. The abnormality is not anatomically suggestive of a parenchymal abnormality and has vessels seen to be coursing through it, further supporting this is not likely due to a parenchymal air space opacity. There is no blunting of the right costophrenic angle to suggest this is loculated pleural fluid elsewhere. The left hemithorax is clear and the cardiomediastinum a little distorted by a mild scoliosis but otherwise unremarkable. It is noted that there are old posterior fractures of the right ninth and tenth ribs.
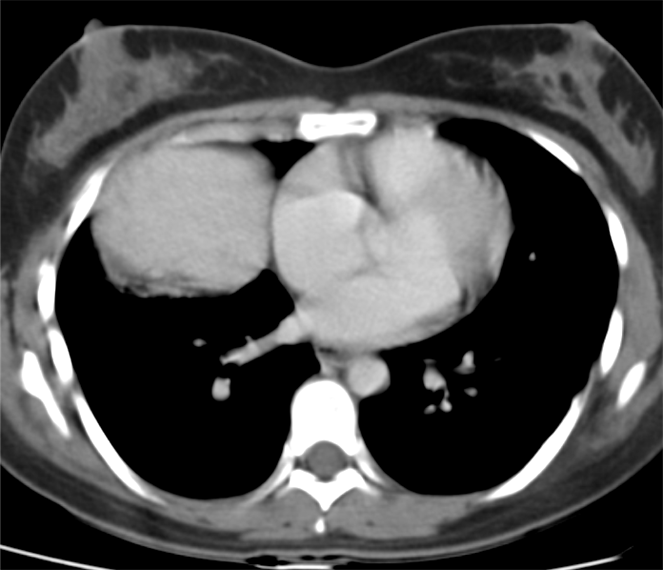
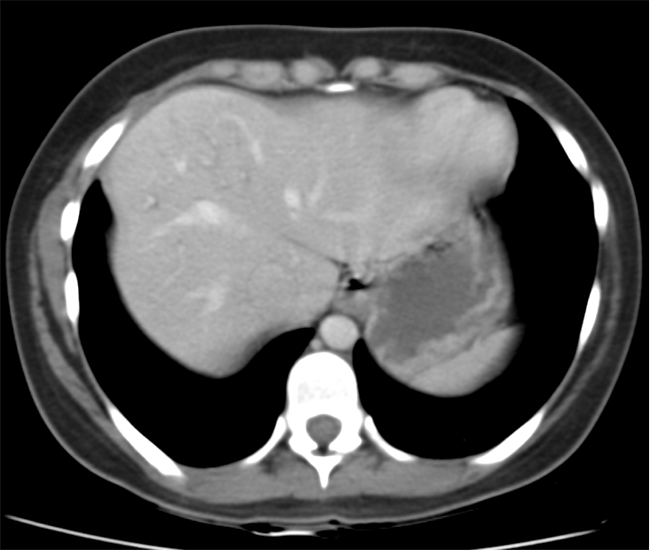
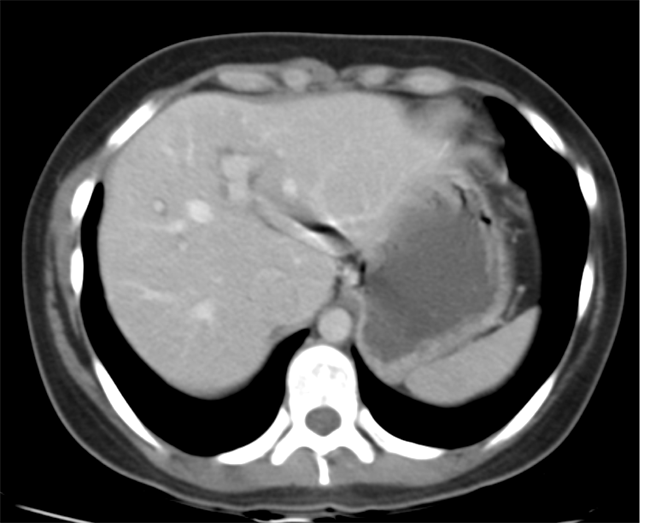
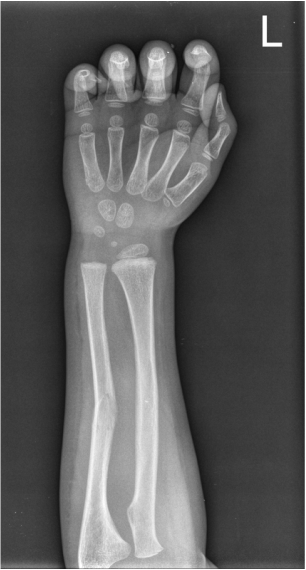
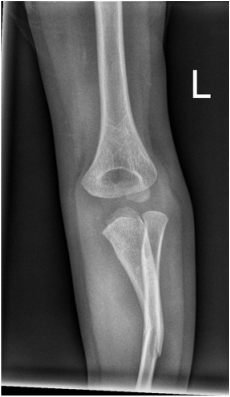
The CT examination is illuminating. This demonstrates no overt pleuroparenchymal abnormality. There is elevation of the anterior aspect of the right liver lobe with a notched appearance to the lateral margin of the liver on the bottom left image. Also noted is that there is low attenuation along the vessels in the anterior segment of liver. This suggests that there is segmental mild biliary obstruction in this region, but not centrally in the porta hepatis. The remainder of the right posterior liver and left liver lobe are unremarkable. The right lateral margin of the liver in contact with the right lateral chest wall has lost its normal rounded configuration and has a squared off appearance. These appearances are indicative of a right diaphragmatic traumatic injury with partial herniation of the liver through the diaphragmatic tear. This results in the notch in the lateral liver aspect as well as the segmental ductal obstruction of the herniating segments. Diaphragmatic tearss are not uncommonly delayed presentations following trauma. In part this may because patients with trauma may be treated with positive pressure ventilation which may suppress a diaphragmatic injury. But also tears can enlarge with time becoming sympomatic. Diaphragmatic injuries are commoner on the left side and less so on the right side, thought to be due to the protective of the liver. Most commonly these are due to increased intra-abdominal pressure from blunt injury but can also of course occur with penetrating injuries. Delayed diagnosis results in increased morbidity and mortality, particularly as larger hernias can become more difficult to surgically repair. CT is essential in confirming the diagnosis of diaphragmatic tears, particularly the coronal and sagittal reconstructions, although these are less helpful on the right side due to close approximation of the liver to the diaphragmatic fibres, frequently with no substantial interposing fat. This case was diagnosed based on the abnormal configuration of the right diaphragm with evidence of prior trauma (rib fractures), aided by the focal biliary tract obstruction. Such cases should be differentiated from diaphragmatic eventration which is due to an anteromedial weakness of the diaphragm. In this context the PA chest x-ray would demonstrate loss of the entirety of the right cardiac border near the elevated segment and there would be no evidence of biliary obstruction present. Case Level: Radiology Junior +, ABR, EDiR, MRCP, FRCR, FRCS The images demonstrate a relatively obvious fracture of the ulna. This is appreciable on the AP projection and confirmed as significantly angulated on the lateral projection. However, careful review of the elbow image reveals an additional abnormality. What is it?
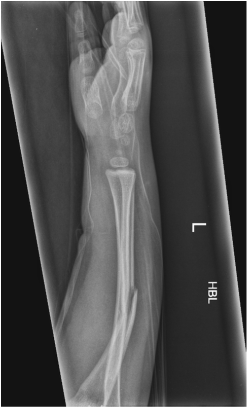
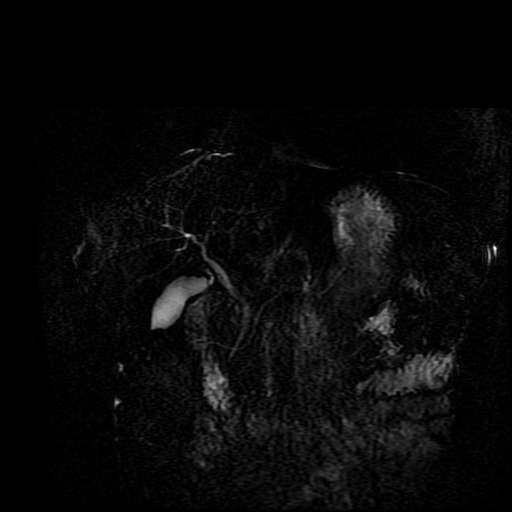
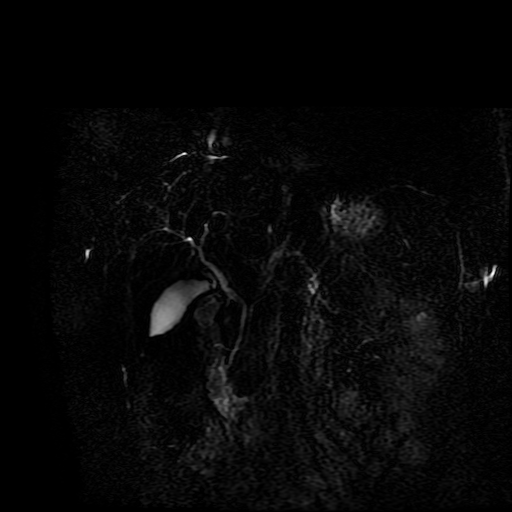
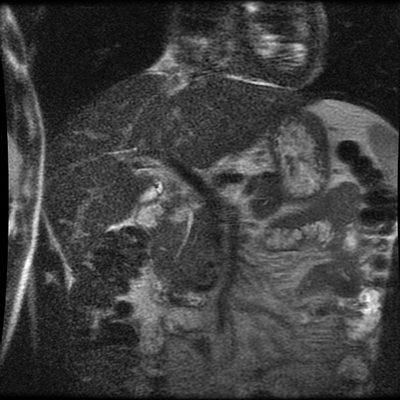
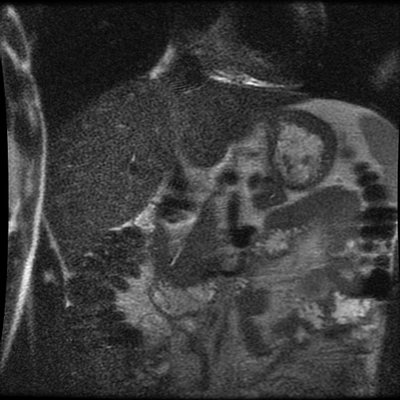
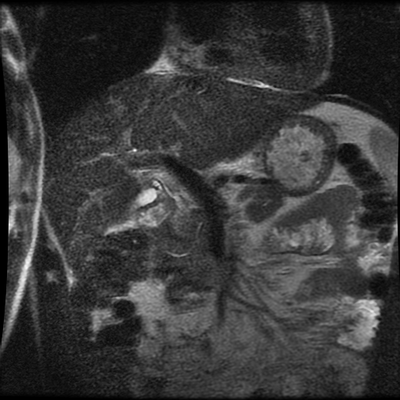
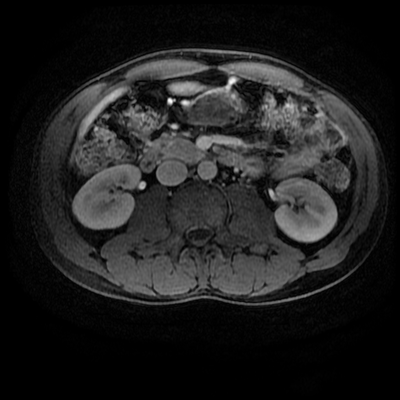
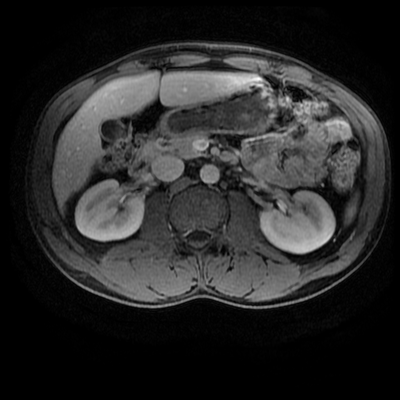
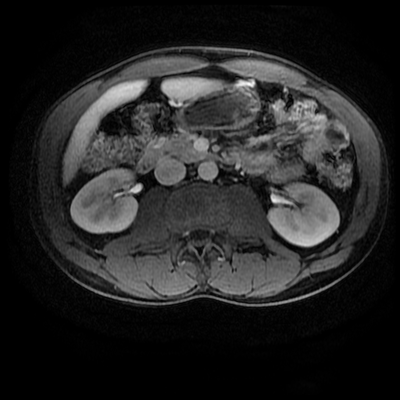
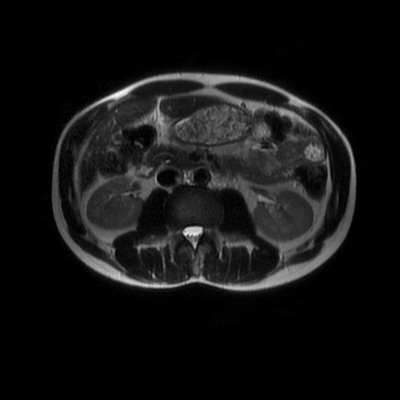
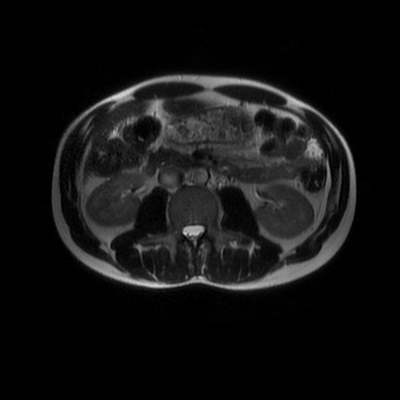
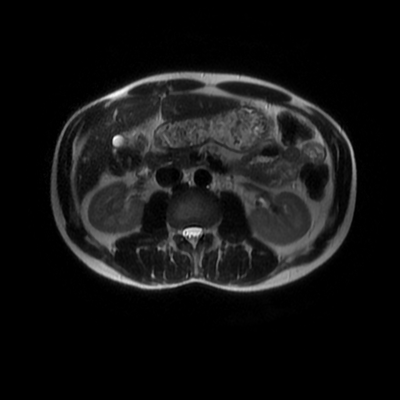
Whenever reviewing a paediatric elbow the first consideration is to establish that all the expected epiphyses are present. These appear in the order defined by the mnemonic of CRITOL (Capitellum, Radial head, Internal epicondyle, Trochlea, Olecranon, Lateral epicondyle). Epiphyses appearing out of order can be due to avulsion-dislocation of an epiphysis. In this case only the first epiphysis, the capitellum, in its correct position. However, the capitellum is the “cap” that lies over the radial head. In this case we can see that a line drawn through the medulla of the radius, through its expected head location does not meet the capitellum. Therefore, the findings reflect dislocation of the proximal radius/radial head. The combination of a mid-ulnar fracture and proximal radius/radial head dislocation constitutes the eponymous fracture-dislocation combination of Monteggia. This type of fracture dislocation highlights the importance of imaging the joint above and below a fracture. These types of fracture-dislocations are difficult for most to remember. The commonly confused fracture-dislocation is that of Galeazzi. In Galeazzi there is a mid-radial fracture with dislocation of the distal ulnar head at the wrist joint. It can be frustrating to remember the fracture and where the dislocation is (bone and location) and there are many mnemonics to assist. My preference is to recall that the dislocation occurs in the non-fractured bone and to use the mnemonic of GRIMUS (Galeazzi, Radius #, Inferior dislocation, Monteggia, Ulna #, Superior dislocation). Recall that the fracture may simply be a greenstick injury but it is essential to look for the associated dislocation as these can be missed and result in chronic pain/dysfunction for the patient and medicolegal pain for the reporting radiologist! Finally don’t forget the final fracture dislocation which is the Essex-Lopresti (one hyphenated British surgeon not 2 people!). This is a comminuted fracture of the radial head where force is transmitted down the interosseous ligament resulting in dissociation of the distal radio-ulnar joint at the wrist. Level: MRCP, FRCR, FRCS, EDiR, Radiology Mid-Level ++ The images provided part of an MRCP acquisition. The two superior images are radial ray thick slab heavily T2-weighted acquisitions (oblique views). The second series of images are coronal thin section T2 weighted single shot fast spin-echo (SSFSE sequences). The third row of images are T1 fat suppressed volumetric acquisitions following intravenous gadolinium (LAVA, equivalent to VIBE or THRIVE). The final row of images are T2 axial images.
The images demonstrate that there is a crossover of the main pancreatic duct and of the distal common bile duct. The main pancreatic duct drains into the the accessory duct (Santorini) which emerges into the duodenum from the proximal minor papilla. The uncinate process of the pancreas drains via the main pancreatic duct (Wirsung) into the main ampulla, also receiving the common bile duct. These appearances are those of pancreas divisum. This appears due to failure of fusion of the ventral pancreatic bud with the larger dorsal pancreatic bud in the 8th week in utero. The ventral pancreas drains through the Wirsung duct which after the 8th week typically becomes the distal main pancreatic duct emerging into the duodenum at the ampulla. The duct of Santorini which drains the dorsal pancreas distally into the minor ampulla usually becomes atrophic allowing drainage through the more inferior Wirsung duct. Note that confusingly due to rotation of the pancreas in utero the posterior inferior head of the pancreas is actually congenitally the ventral pancreas. When the two duct systems fail to fuse pancreas divisum arises. Pancreas divisum is present in approximately 10% of patients, depending on how this is evaluated (postmortem 7%, ERCP 10-14%) and the definition of divisum. Although Asian populations have been reported to have lower incidence (1-2%) this has been stated in part in part to relate to less invasive testing. Many of the higher incidences of pancreas divisum relate to a definition that includes predominant flow into the duct of Santorini with small branch duct communication from the pancreatic duct to a small Wirsung duct. Radiologically the term at MRCP is usually confined to cases where there is complete separation with no bridging duct between the pancreatic duct in the neck/head and the Wirsung duct. This results in a crossover appearance of the CBD extending down into the ampulla and of the pancreatic duct extending into the duct of Santorini. Most cases of pancreas divisum are asymptomatic. In a small minority of cases recurrent pancreatitis may be related to pancreas divisum, due to inadequate drainage from the minor ampulla and the narrowed distal duct of Santorini. This can result in focal ovoid dilatation proximal to the ampulla (Santorinicoele). In these cases treatment can be by sphincterotomy or duct stenting. |
From Grayscale
Latest news about Grayscale Courses, Cases to Ponder and other info Categories
All
Archives
October 2018
|

|
|
Grayscale Courses est. 2015
 RSS Feed
RSS Feed
